Introduction
Functional gastrointestinal disorders (FGIDs) are very common diseases [1]. Most gastrointestinal (GI) disorders are diagnosed from a physiological abnormality or biomarker, and no defintive pathophysiological features characterize FGIDs [2,3]. Therefore, it is necessary to exclude other diseases before a functional GI designation can be made [4].
The Rome III criteria were updated to identify a more homogeneous patient group for pathophysiological studies and clinical trials [4,5]. There have been some changes to the definitions of FGIDs according to the ROME III criteria [5,6]. The first is a time-frame change for FGIDs: symptoms must have begun 6 months prior to diagnosis and be active for recent 3 months. This time frame is less restrictive than that of Rome II criteria (12 weeks of symptoms over a 12 months perioid). The second change in the criteria for functional dyspepsia (FD). FD is divided into the two subcategories of postprandial distress syndrome (PDS) and epigastric pain syndrome (EPS), which are similar to the dysmotility-like and ulcer-like dyspepsia, respectively, of the Rome II criteria. The third change is a revision of the irritable bowel syndrome (IBS) subtypes: diarrhea, constipation, and the mixed and unspecified subtypes should be based on a simple classification derived from stool consistency.
Talley’s bowel disease questionnaire (BDQ), a self-reported structured questionnaire, has been used to measure the symptoms of FGIDs and past medical histories. It is reliable, with a median kappa value of 0.78 (range, 0.52∼1.00), and adequate content and construct validity [7,8]. The BDQ contains 46 GI-related items and an additional 14 questions in drop-down boxes. It includes 26 items about GI symptoms, medical visits, sociodemographic data, body mass index, family history, and surgical history.
The responses to the questionnaire, including those about GI symptoms, might be influenced by the social and cultural milieux. Therefore, a simple translation of the questionnaire from English to another local language is insufficient because the cultures and conceptual uses of language will differ.
In the present study, we developed a Korean version of the BDQ (BDQ-K), using a standard translation process, and validated it. We also investigated whether the symptom criteria distinguished the patients presumed to have IBS (by a positive diagnosis) from healthy subjects or patients with other functional or structural disorders.
Methods
We used the modified version of the BDQ. Because the symptom items of the BDQ were tested individually, subsequent questionnaires have used selected questions from the original instrument. A new questionnaire, the BDQ-K, was developed using validated questions from previous surveys and new questions, incorporating all symptom items required to meet the ROME III criteria for the chief complaint. The BDQ-K contains 31 GI-related items and an additional 14 questions in drop-down boxes. It includes 11 items referring to GI symptoms, medical visits, sociodemographic data, body mass index, family history, and surgical history. The patients were given a diagram of the abdomen and asked to mark the site of any abdominal pain, to facilitate a classification of upper- and lower-abdominal pain. The somatic symptom checklist consists of 18 non-GI symptoms or illnesses. The total length of the instrument is 12 pages.
The BDQ-K was reviewed by elementary school teachers to ensure that the terminology was comprehensible at a sixth-grade reading level. The questionnaire was translated into Korean by a bilingual nonmedical person. Back-translation was performed by another bilingual nonmedical university student. The investigators reviewed the translated and back-translated questionnaires to ensure that the relevant GI symptoms could be properly assessed. For pretesting, the BDQ-K was given to 10 consecutive outpatients in the gastroenterology outpatient clinic in Ewha Womans University Mokdong Hospital, who were asked to complete the survey and comment on any questions that were difficult to understand.
The reliability of the BDQ-K was determined with a test-retest study on 69 independent, consecutive outpatients. The patients completed an identical BDQ-K a second time by mail within 4 weeks of completing an initial questionnaire. The outpatients volunteered to participate as the independent sample in the validity test of the two versions of the BDQ-K, with a sex-stratified random order. Two doctors who were blinded to the prior medical histories and responses of the patients interviewed them and subsequently completed the BDQ-K based on the information obtained during the interviews.
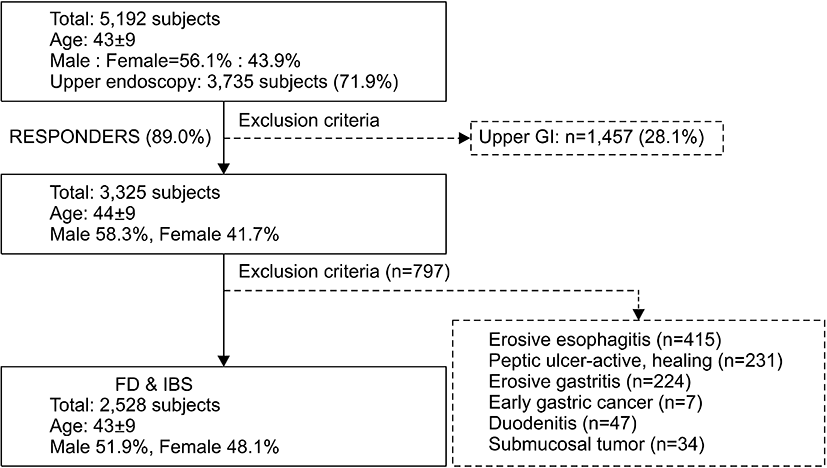
A total of approximately 5,192 subjects at a Health Promotion Center at Ewha Womans University Mokdong Hospital who were over 18 years of age were enrolled from August 2008 to January 2009. Of these, 1,457 subjects (28.1%) were excluded because they had undergone radiologic upper-GI series rather than endoscopy (Fig. 1).
In total, 3,325 subjects (mean age, 44±9 years; 58.3% male) presenting for 3,735 endoscopies responded to the BDQ-K, generating an 89.0% response rate. The exclusion criteria were: 1) no upper endoscopy; 2) could not read Korean; 3) could not understand the BDQ-K because of severe alcoholism, dementia, old age, etc.; and 4) refusal to participate. Among the 3,325 subjects, 797 were excluded because of organic diseases, including erosive esophagitis (n=415), active or healing stages of peptic ulcer (n=231), erosive gastritis (n=224), early gastric cancer (n=7), duodenitis (n=47), and submucosal tumor (n=34). Finally, a total of 2,528 subjects who responded to the questionnaire and underwent an upper endoscopy were included in the study. The study protocol was approved by the Institutional Review Board of Ewha Womans University Mokdong Hospital.
FD was defined according to the ROME III criteria [5,6]: (1) PDS, or (2) EPS in the current 3 months and no evidence of structural disease (including on upper endoscopy). IBS was defined as recurrent abdominal pain or discomfort, with no evidence of structural disease, plus two out of three of the following symptoms [6]; (1) pain relief after defecation, (2) pain onset associated with a change in stool frequency, (3) pain onset associated with a change in the stool form (appearance) during the current three months. IBS was classified into four types: constipation predominant, diarrhea predominant, mixed, and unclassified.
The statistical analysis was performed using the kappa statistic for reliability and validity. A kappa statistic value of 0.4∼0.7 indicated a good correlation, and a value of >0.7 indicated an excellent correlation [9]. Continuous variables were expressed as means±SD. We performed a χ2 test for the prevalence of sex-based differences. Data analyses were performed with SPSS for Windows (v. 13.0; SPSS Inc., Chicago, IL, USA), and P<0.05 was considered significant.
Results
The median kappa value for the test-retest study was 0.56 (range 0.22∼1.0) for the GI symptoms; κ=0.51 for abdominal pain (P<0.001), κ=0.51 for pain onset before six months (P<0.001), κ=0.64 for pain severity (P<0.001), κ=0.69 for epigastric pain (P<0.001), κ=0.40 for early satiety (P<0.001), and κ=0.34 for postprandial fullness (P<0.001; Table 1).
In the validity study of the BDQ-K, the median kappa value was 0.74 (0.36∼1.0); κ=0.91 for abdominal pain (P<0.001), κ=0.71 for pain onset before six months (P<0.001), κ=0.83 for pain severity (P<0.001), κ=0.73 for epigastric pain (P<0.001), κ=0.50 for early satiety (P<0.001), and κ=0.57 for postprandial fullness (P< 0.001; Table 1).
For the non-GI symptoms, the median kappa value of the test-retest study was 0.63 (range 0.27∼1.0), including κ=0.66 for weight loss (P<0.001), κ=0.81 for surgical history (P<0.001), κ=0.80 for pregnancy history (P<0.001), and κ=0.75 for frequency of visits to doctors (P<0.001). The median kappa value of validity was 0.76 (range 0.36∼1.0; Table 2).
A total of 2,528 subjects (mean age, 43±9 years; 51.9% male) were included in the study. The prevalence of FD at least weekly was 8.3% (209/2,528). A female predominance was noted in the subjects with FD (M:F=10.8:5.9, P<0.001; Table 3).
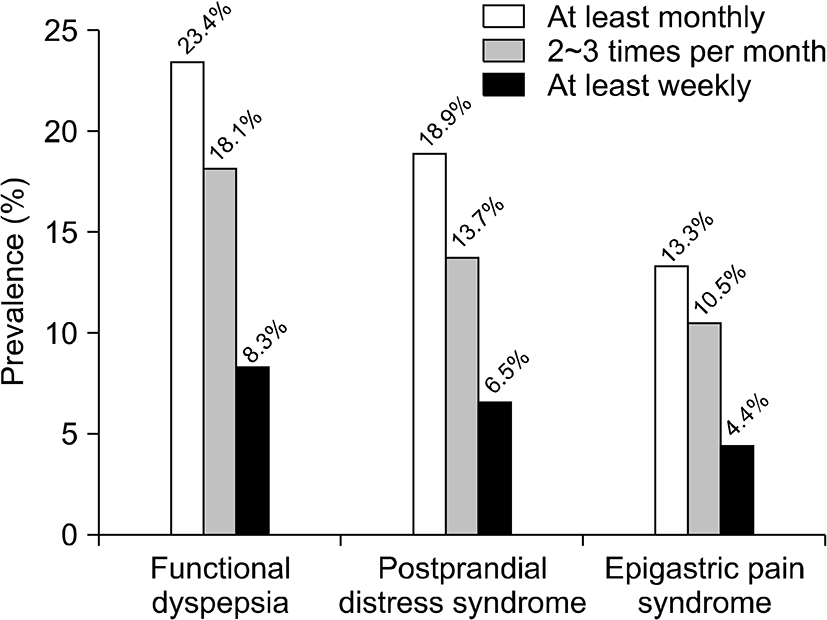
The proportion of patients with EPS was 4.4% (112/2,528) and with PDS was 6.5% (164/2,528), which consisted of early satiety 2.5% (64/2,528) and postprandial fullness 6.1% (155/2,528; Fig. 2).
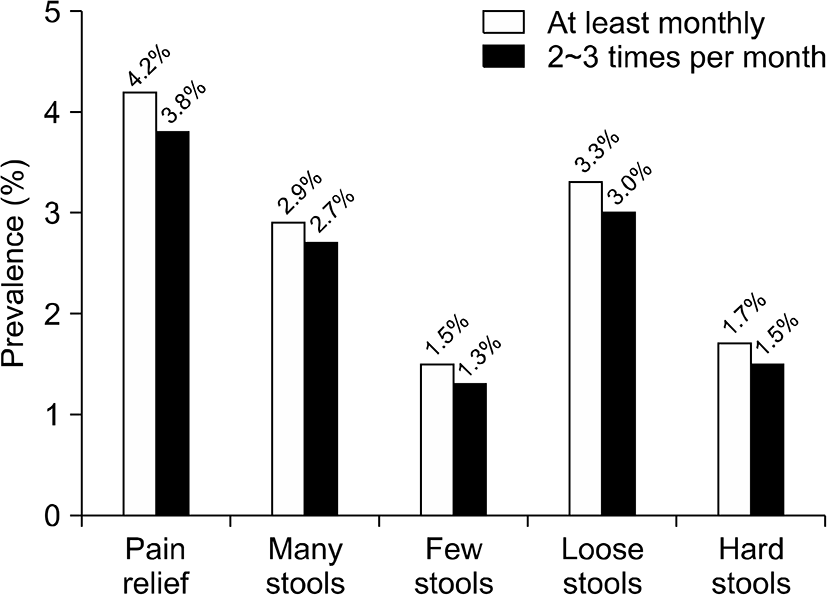
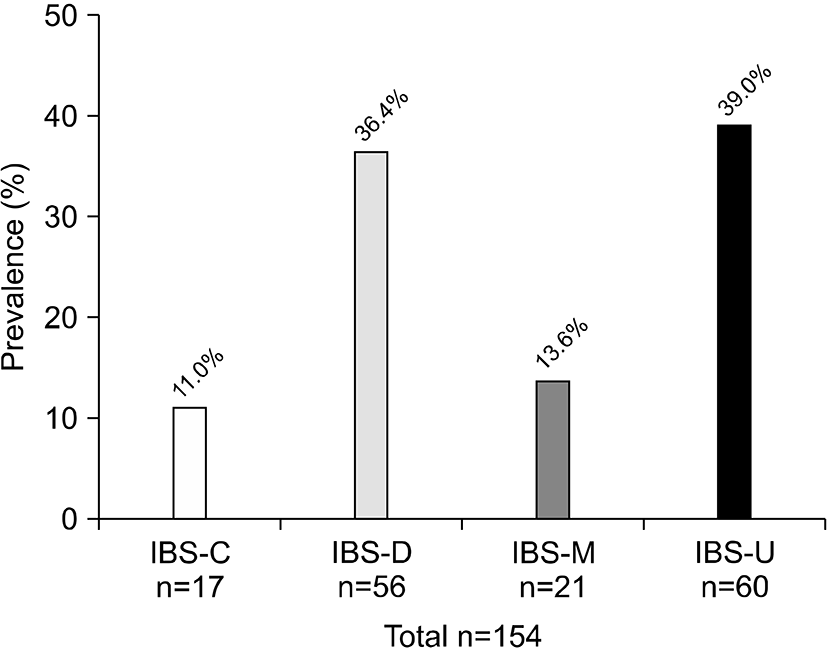
The prevalence of IBS at least 2∼3 times per month was 6.1% (154/2,528) and at least once a month was 6.8% (171/2,528). IBS also predominated in women (7.1% vs 5.1% in men, P=0.032; Table 4). According to the proportion of lower-abdominal symptoms, pain or discomfort improved after a bowel movement at least 2∼3 times per month in 3.8% of patients; the onset of pain or discomfort was associated with more stools at least 2∼3 times per month in 2.7%; fewer stools occurred in 1.3%; looser stools occurred in 3.0%; and harder stools occurred in 1.5% (Fig. 3). Among the 154 subjects who had IBS symptoms at least 2∼ times per month, the prevalence of the IBS subtypes were as follows: IBS constipation 11.0% (n=17), IBS diarrhea 36.4% (n=56), IBS mixed 13.6% (n=21), and IBS unspecified 39.0% (n=60; Fig. 4).
Discussion
FGIDs are highly prevalent and potentially disabling conditions that are diagnosed by the patterns of symptoms presented by the patients to their physicians. For the purpose of research, we need a reliable and standard method to properly select patients to analyze the specific pathophysiological features underlying these symptoms, and to develop proper treatments for targeted patient groups. However, it is difficult to validate the symptom-based diagnostic criteria for FGIDs because of the lack of gold standard diagnoses. In the absence of a gold standard diagnostic factor or biomarker, investigators have adopted one of the following two strategies. One defines FGIDs by exclusion and the other defines FGIDs by positive symptoms.
Our study is the first to translate into Korean the BDQ, a well-known validated, structured questionnaire for the evaluation of FGIDs, and to validate it with a standardized method. The questionnaire, which is based on self-reported symptoms, can guide diagnoses and treatment approaches. It may also facilitate objective diagnosis and increase their reliability, and reduce the ordering of unnecessary diagnostic studies for FGIDs. It may also be important in socioeconomic context to evaluate different treatment outcomes, and in the establishment of dynamic clinical changes. Therefore, a well designed verified questionnaire may be helpful for diagnosing FGIDs.
In Korea, there are several questionnaires for the diagnosis of FGIDs, which have been translated or specifically developed [10,11]. Cho et al [12] translated into Koreans the questionnaire developed by Locke [13] for gastroesophageal reflux disease (GERD), thus generating the GERD-K. The GERD-K includes a questionnaire about GERD and a medical outcome study, the short form 20 (SF-20), a simplified questionnaire of the patient’s quality of life. Questionnaires about GERD, FD, IBS, and functional constipation defined according to the ROME II criteria [13,14] have also been translated into Korean [15,16]. A few studies [17,18] of the epidemiology of IBS based on one of these translated questionnaires have also been undertaken, according to the ROME II criteria [19]. The validity and reliability of the questionnaire in assessing the quality of life of IBS patients were also documented [20].
Epidemiological studies in Korea have demonstrated the prevalence of FD and IBS, defined according to the ROME II criteria, to be around 9.5∼12.2% and 6.6∼9.6%, respectively [15-18]. However, information on the prevalence of FD and IBS in Asia is still lacking, especially for Korea. In the past, a factor analysis of the symptoms in a sample of Asian patients with FGID supported the use of the ROME II classification [21]. In subject undergoing a health check-up, the frequency of FD was 13.4% based on the ROME II criteria [22]. Another study showed that the prevalence of IBS, defined according to the ROME II criteria, was 16.8% [23]. Therefore, the results differ among studies and the response rates were two low to represent the Korean population. In our study, the prevalence of FD and IBS, defined according to the Rome III criteria, was 8.3% and 6.1%, respectively.
In the present study, the accuracy of the BDQ-K was excellent, with a median kappa value of 0.74 (0.36∼1.00). However, its reliability was relatively low (κ=0.56, range 0.22∼1.00). The median follow-up duration was two weeks after the first visit. However, GI symptoms fluctuate within several weeks and sometimes disappear after an endoscopy or clinical work-up for GI symptoms. Therefore, a two-week interval for the test-retest study might have been too long for the evaluation of FGIDs.
The lowest kappa value was observed for upper-GI symptoms, especially early satiety, postprandial fullness, and hunger pains. In the present study, the prevalence of early satiety was relatively low, but was not proportionally low as that for postprandial fullness or epigastric pain. It is difficult to say whether this result represents true poor quality of questionnaire or the different socio-cultural effects experienced in Korea compared with Western countries.
The subgroup classification of IBS is important for clinical trials, especially pharmacodynamic studies. The Rome III criteria recommend that the classification of IBS subtypes by the Bristol Stool Scale (BSS). However, the response rate to the BSS was very low and Korean subjects did not readily understand it. There are few data available about the duration or frequency of stool form evaluated with the BSS. We used the BSS questionnaire to evaluate the last bowel movement according to the original BDQ, but, many subjects did not remember it. Consequently, we failed to assess the reliability of the BSS. Therefore, the subtypes of IBS were not easily approached. We used this analysis following the specific questionnaire for stool form in the Rome III questionnaire on the homepage of Rome committee [24].
This study has several strong points. First, we enrolled a large sample of study participants who underwent laboratory studies, upper endoscopy, and abdominal ultrasonography. Second, the response rate to the questionnaire in this study was very high, at 89.0%. The response rates have been reported to be <50% in several recent studies [15,18]. The study also has some limitations. The kappa statistic was relatively low for the reliability of the questionnaire and this was mainly related to the misunderstanding of symptoms that commenced six months earlier. Some kappa statistics could not be measured because of the inconsistency of several different answers. We enrolled relatively large numbers of subjects in the study, but most of them did not have FGIDs, so the low rate of positive symptoms might have influenced the agreement in the kappa statistics. A proportion of the subjects who were enrolled at the Health Promotion Center had different exposures from general population.
In conclusion, the BDQ-K is an accurate and reliable instrument for identifying FGIDs according to the Rome III criteria. The prevalence of FD and IBS according to the Rome III criteria was 8.3% and 6.1%, respectively.