Introduction
Herpes simplex virus (HSV) infection is a common and usually benign, self-limiting disease, which normally presents with mucocutaneous lesions and mild viremia, although the primary HSV infection can cause rather severe local infection [1].
Occasionally, HSV is associated with severe life-threatening or fatal infection by disseminating to visceral organs. Herpes simplex hepatitis is a well-known clinical condition described in the pediatric population and pregnant women. In adults, systemic herpes simplex with acute hepatitis is a rare complication of HSV infection. It occurs in two distinct adult populations: immunocompromised adults and pregnant women in the late second or third trimester [2,3].
Early diagnosis may be difficult when the characteristic vesicular rash is absent, early symptoms are often nonspecific, and there is a lack of history of genital herpes infection [4,5]. Here, we present a female patient with hepatitis induced by HSV. We initiated intravenous (IV) acyclovir at a high dose early in the course of the disease. Rapid initiation of IV acyclovir enabled successful treatment, and the patient recovered well.
Case
A 33-year-old woman was admitted to the hospital owing to right upper quadrant abdominal pain and general weakness for three days. The patient had a medical history of breast cancer 10 years ago and a 10-year history of diabetes mellitus, for which she had been taking oral anti-hyperglycemic medications.
On admission, vital signs were as following: blood pressure, 115/86 mmHg; heart rate, 131 beats/min; respiratory rate, 20 breaths/min; and body temperature, 37.5°C. On physical examination, the patient had an acute ill-looking appearance and presented normal conjunctivae. On abdominal examination during admission, the patient had normoactive bowel sounds without tenderness or rebound tenderness. The patient’s head and neck examination was unremarkable, and no oral lesions were documented.
Laboratory measures were as following: hemoglobin, 13.5 g/dL; hematocrit, 40.5%; white blood count, 9,660/mm3; platelet count, 277×103/mm3; sodium, 137 mEq/L; potassium, 4.6 mEq/L; chloride, 99 mEq/L; blood urea nitrogen, 30.9 mg/dL; serum creatinine, 1.4 mg/dL; aspartate aminotransferase (AST), 3,455 IU/L; alanine transaminase (ALT), 2,970 IU/L; alkaline phosphatase, 639 U/L; total bilirubin, 3.0 mg/dL; international normalized ratio, 1.403; prothrombin time, 52%; partial thromboplastin time, 30.0 seconds; amylase, 49 IU/L; and lipase, 26 U/L.
The patient had no history of high-risk behaviors such as alcohol abuse, smoking, illicit drug use, or indiscriminate sexual behaviors. She had no dermatological, obstetric, or gynecologic findings such as herpes, wart virus, genital infection, or candidiasis. As treatment for suspected hepatitis, the patient was prescribed flavine adenine dinucleotide sodium at 20 mg/day for 10 days.
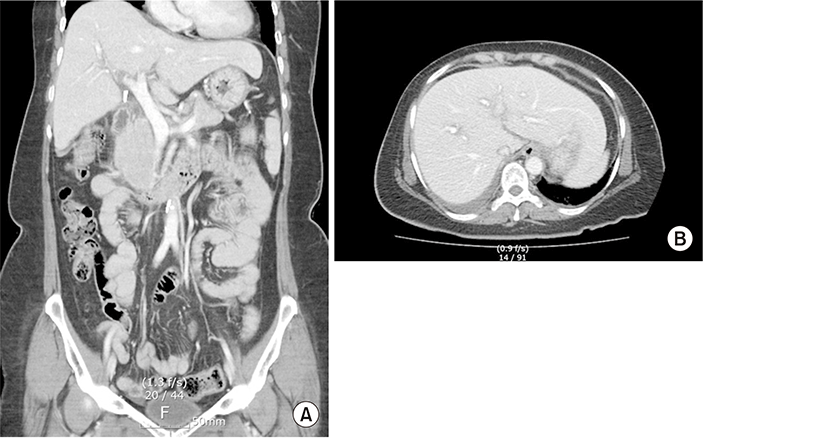
On day 2, serologic test results were hepatitis B surface antigen nonreactive and surface antibody reactive, hepatitis A IgG antibody nonreactive and IgM antibody nonreactive, and hepatitis C antibody nonreactive. Doppler ultrasound of the liver was remarkable for heterogeneous echotexture of the liver as well as blood flow in the normal direction in the major hepatic vessels (Fig. 1). A computed tomography scan of the patient’s abdomen with a contrast agent revealed a small amount of ascites, diffuse heterogeneous enhancement of the liver with a few tiny small nodules, and no further known cancer progression (Fig. 2).
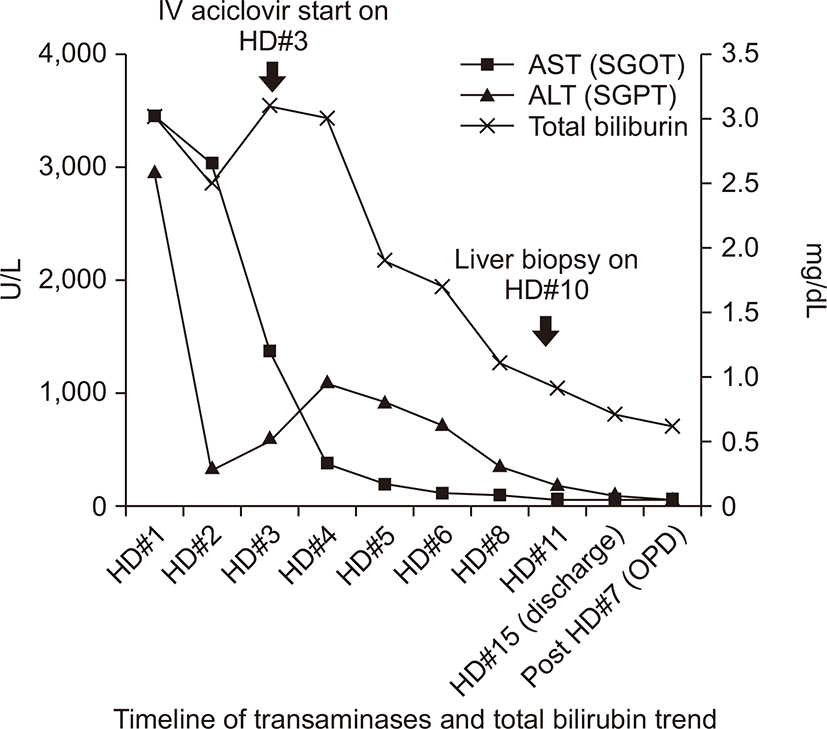
On day 3, immediately after obtaining the above results, the patient was screened for multiple additional viruses, including parvovirus B19, cytomegalovirus, Epstein-Barr virus, all of which were negative; however, the test for HSV IgM antibody was positive. The patient was immediately started on IV acyclovir (10 mg/kg IV at every 8 hour for 5–10 days).
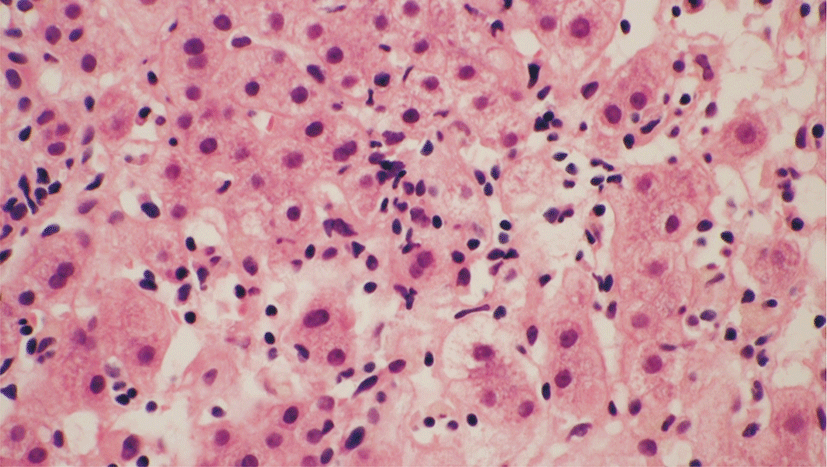
On day 6, the AST level decreased significantly to 1,350 IU/L. On day 10, a percutaneous liver biopsy was performed because of the patients’ poor condition and prolonged prothrombin time on admission. Examination of biopsy specimens revealed reactive changes of hepatocytes and lobular necrosis associated with mild cholestasis. Immunostaining for HSV was nonspecific, and the results of viral cultures were negative (Fig. 3).
Although her liver biopsy did not reveal HSV, she completed a 2-week course of IV acyclovir treatment and survived. She was discharged without further treatment and did not experience any recurrence during follow-up in our outpatient department (Fig. 4).
Discussion
HSV hepatitis is rare and accounts for only 1% of all acute liver failure cases and only 2% of all viral causes of acute liver failure [4-6]. Several risk factors are associated with the development of HSV hepatitis, including burns, cancer, pregnancy, or immune-modulation drugs: the similarity in each of these conditions is that the host immune defenses are compromised [7]. As HSV hepatitis is associated with high mortality, exceeding 80%–90%, it seems reasonable to urge early aggressive diagnosis and antiviral therapy, even in the absence of characteristic mucocutaneous ulcerative lesions [8].
Diagnosis is often difficult to establish because of the lack of specific symptoms and the absence of typical herpetic lesions, as was the case in the patient. Clinical manifestations are nonspecific acute symptoms, which include flu-like illness, fever, and abdominal discomfort with rapid progression to hepatic necrosis and coma [1,5]. Laboratory investigations often show leukopenia, thrombocytopenia, and coagulopathy. Renal failure occurs in up to 65% of patients with HSV-related acute liver failure. Moreover, disseminated intravascular coagulopathy is a late sign of the disease. Ninety percent of patients with HSV hepatitis have a characteristic liver profile, known as “anicteric hepatitis,” which refers to a liver profile showing a significant increase in transaminases (100–1,000 fold) with relatively normal or low bilirubin [4]. There may be a marked elevation of AST, greater than that of ALT [4,5].
Prompt diagnosis of non-acetaminophen fulminant hepatic failure should not only include studies of common hepatitis viruses such as hepatitis A and B, Epstein-Barr virus, and cytomegalovirus, but also HSV [4].
Seventy-two percent of patients have identifiable mucocutaneous lesions, yet herpes hepatitis was only considered in the differential diagnosis in up to 33% of patients [8]. These lesions should be subjected to smear examination for giant cells and culture tests for viruses. Liver biopsy should be undertaken without delay because antiviral therapy changes may result in loss of typical viral inclusion bodies [8]. Liver biopsy is the gold standard diagnostic method, but owing to the low level of suspicion and the high risk of bleeding caused by prolonged prothrombin time, it was performed in only one-third of the patients [9]. However, the value of liver biopsy may help in confirming suspected HSV hepatitis.
The current patient received early empirical treatment with acyclovir with HSV hepatitis. This case clearly illustrates how prompt therapy can positively affect the hospital course of the patient. The administration of IV acyclovir is inexpensive, has no drug interactions, and is safe even during pregnancy and before liver biopsy.
Percutaneous liver biopsy in the current case did not demonstrate HSV; this explains the missed diagnosis, presumably because of the efficacy of acyclovir in eradicating HSV from hepatocytes. On the other hand, the negative immunostaining results also could be attributed to sampling error because of the small sample size obtained with a needle biopsy.
The present case describes a non-immunocompromised, non-gravid adult patient who was diagnosed with HSV hepatitis. Although HSV hepatitis is associated with high mortality, early initiation of acyclovir might have been lifesaving in this case. Accordingly, HSV must be considered in all patients presenting with liver failure of unknown cause. If suspected, prompt treatment with acyclovir should be initiated early and rapidly.