Introduction
With the increase of immunocompromized or severely ill patients with diabetes or hematologic malignancies, the incidence of invasive fungal infections is on the rise globally. Although new antifungal agents have become available, invasive fungal infections continue to be significant sources of morbidity and mortality in hospitalized patients [1]. Poor outcome of invasive fungal infections is not only related to patients’ impaired immunity or comorbidities but also lack of rapid and sensitive diagnostic modalities, and the resulting delayed initiation of treatment [2]. Invasive aspergillosis is the principal form of invasive fungal infection, with most cases caused by Aspergillus fulmigatus. However, other Aspergillus species and Zygomycetes, such as mucorales, have recently emerged as important invasive fungal pathogens [3].
It is crucial to distinguish invasive aspergillosis and mucormycosis because these two mold infections have different susceptibilities to antifungal agents. However, diagnosis of an invasive fungal infection can be challenging since current diagnostic methods are not sufficiently sensitive or specific, and require an extended time that is clinically prohibitive. Radiologically, halo or reverse halo sign on chest computed tomography (CT) scan could aid differential diagnosis. Studies have reported that reverse halo sign favors pulmonary mucormycosis rather than aspergillosis in immunocompromised patients [4,5]. However, those radiologic findings is useful only in the early course of the disease, and data for immunocompetent host is still lacking [6]. Fungal culture can be useful in the identification of a particular pathogen. However, the approach is limited by low sensitivity and a relative prolonged time for results. Although microscopic examination of tissue specimens is considered as a gold standard, it suffers from the limitations of the occurrence of atypical morphologies and similar histopathologic characteristics of different organisms [7]. Galactomannan, which is a component of the cell wall of Aspergillus species, can be a useful surrogate marker for invasive aspergillosis. However, false-positive and false-negative reactions remain problematic [8]. In the face of conflicting findings in the conventional diagnostic modalities, nucleic acid-based examinations can enable a conclusive diagnosis [9].
Here, we present a case of invasive pulmonary aspergillosis that histologically mimicked mucormycosis, in which the confirmatory diagnosis was made using nucleic acid-based methods.
Case
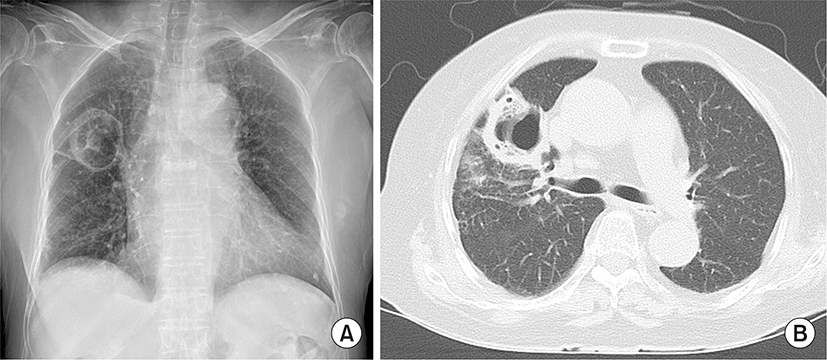
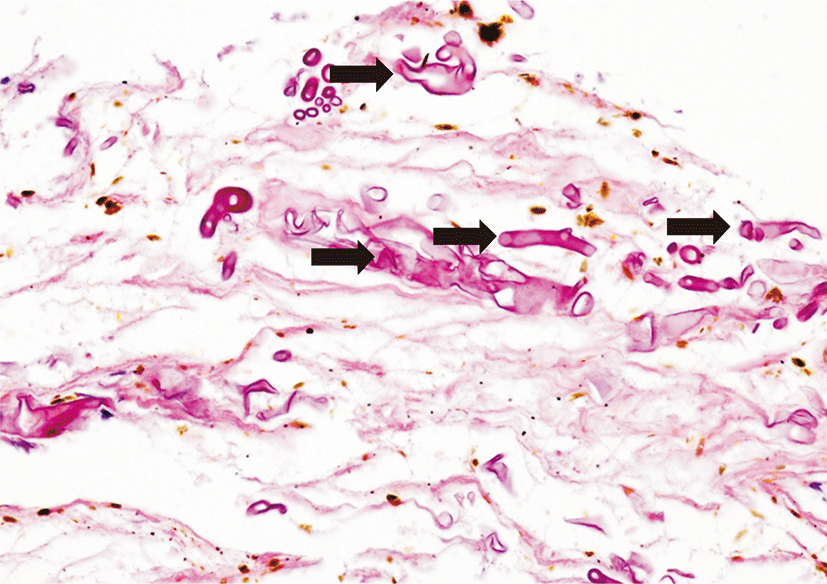
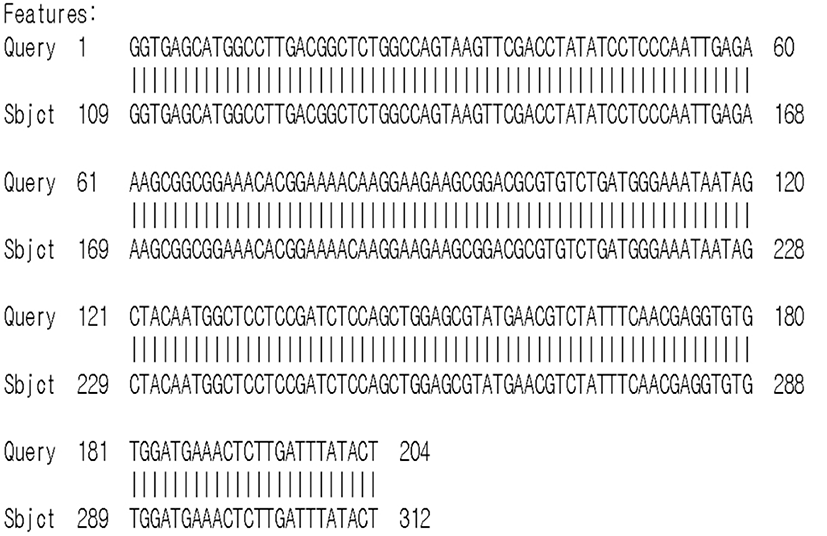
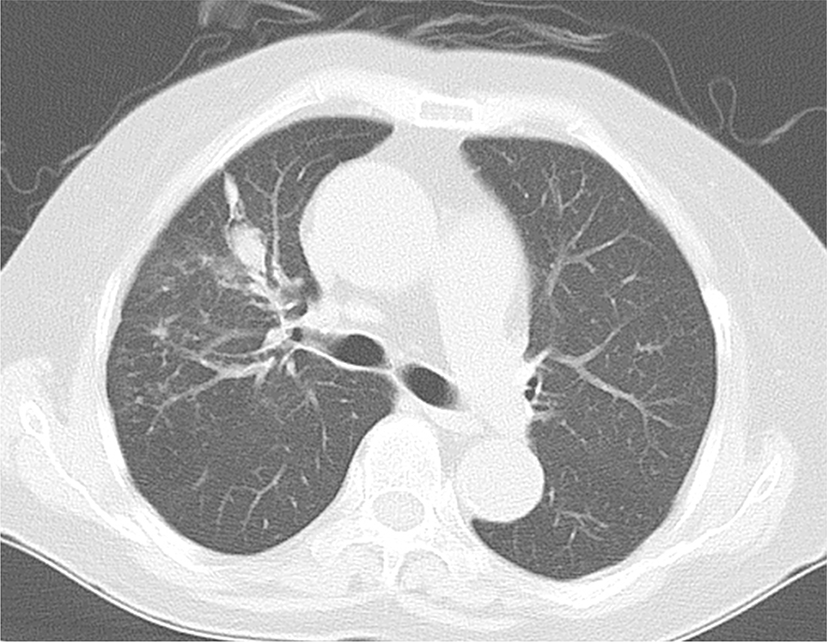
A 75-year-old female presented to the pulmonary department complaining of febrile sensation, cough and sputum for 2 weeks. She had a 15-year history of hypertension, diabetes mellitus, and osteoarthritis, and a 3-year history of adrenal insufficiency due to long-term intake of herb medication. She was taking 2.5 mg of prednisolone per day. Her heart rate was 90 beats per minute, respiration rate was 20 breaths per minute, and body temperature was 37.8°C. Auscultation of the chest revealed no abnormal lung and heart sounds. Laboratory findings revealed a white blood cell count of 12,440/μL (neutrophil 76.4%), C-reactive protein 56 mg/L, and glycated hemoglobin 7.5%. Chest X-ray revealed a cavitary lesion at right upper lobe. Chest CT scan showed a 4 cm-sized irregular-walled cavity with surrounding alveolar consolidation (Fig. 1). Bronchoscopy revealed the absence of endobronchial lesion. Bronchoscopic washing was negative for bacterial and tuberculosis infection. Broad-spectrum antibiotics were administered and CT-guided percutaneous needle biopsy was done under the suspicion of invasive fungal infection. Histopathology revealed broad, non-septated and irregularly-shaped hyphae with infiltration of inflammatory cells that was suspicious of mucormycosis (Fig. 2). Although the galactomannan assay was not performed using bronchoscopic washing fluid, the assay using serum was positive (1.33 index). Fungal culture was negative. The discrepant results prompted fungal polymerase chain reaction (PCR) of a tissue specimen and we requested fungal PCR to other institution. The PCR was performed using DNA sample isolated from paraffin-embedded tissue. Internal transcribed spacer (ITS) and β-tubulin genes were used for sequencing to identify species complex and species within complexes, respectively [10]. The sequences derived from the ITS and β-tubulin regions were compared with sequences available in GenBank by means of the BLAST program (http://blast.ncbi.nlm.nih.gov/Blast.cgi) [10]. The PCR results confirmed the presence of A. fulmigatus (Fig. 3). The final diagnosis of invasive pulmonary aspergillosis prompted voriconazole therapy (intravenous for 2 weeks followed by oral maintenance for 2 months). Symptoms gradually improved and follow-up radiography showed a markedly shrunken cavity lesion (Fig. 4).
Discussion
Aspergillus species and Mucorales are the principal pathogen for invasive fungal infection. Differential diagnosis of this infection is important because each pathogen responds differently to different antifungal agents. However, definite diagnosis of invasive fungal infection is difficult as current diagnostic methods do not have sufficient sensitivity and specificity. A clinical trial on the treatment of aspergillosis established a definite diagnosis of aspergillosis in only 40% of cases [11]. In clinical practice, the characteristic morphology of hyphae in histopathologic examination is considered as the gold standard for the diagnosis of invasive aspergillosis. In contrast to mucormycosis, which appears as broad (6–16 μm), nonseptated hyphae with branches at right angles, aspergillosis is characterized by thin, regular, septated, and acute branching hyphae [12]. However, histopathologic examination is challenging in that other fungal pathogen such as Fusarium and Scedosporium species may not distinguishable from Aspergillus species by hyphal morphology, and nondetection of fungi does not necessarily rule out fungal infection [2]. In addition, the risk of bleeding often prevents an invasive procedure, especially in patients with hematologic malignancies. Furthermore, histopathologic results can be inconsistent with those of other methods. In one study, histopathology and culture results were discordant in 17% of the specimens of invasive fungal infection [13]. Thus, diagnosis of invasive fungal infection should be made cautiously and comprehensively.
New diagnostic modalities for fungal infection have drawn attention for their improved sensitivity and specificity. Galactomannan is a useful surrogate marker for invasive aspergillosis as it is released into the surrounding environment during fungal invasion. A meta-analysis reported that serum galactomannan assay had a 71% and 89% diagnostic sensitivity and specificity, respectively, for invasive aspergillosis [14]. However, false positivity can result from the use of β-lactam antibiotics (such as piperacillin-tazobactam) and the presence of certain other invasive mycoses like Penicillium. False-negativity may result from concomitant antifungal use or low infection burden [8].
Increasing evidence supports the use of fungal PCR as a diagnostic tool for fungal infections. Real-time fungal PCR is more rapid, sensitive, and specific method compared to conventional techniques, and so can enable timely initiation of antifungal therapy. In one study, fungal PCR was superior to culture methods in distinguishing aspergillosis and mucormycosis in histologically positive specimens [15]. The combination of real-time PCR and galactomannan assay can improve the diagnostic sensitivity for invasive aspergillosis in patients with febrile neutropenia [9]. Despite of those proven usefulness, fungal PCR has not been widely adopted in routine clinical practice and only a few institution or labs can perform fungal PCR with universal fungal primers.
As shown in this case, fungal PCR is a useful complementary method that can be crucial in the diagnosis of invasive fungal infection. Clinicians should keep in mind that the differential diagnosis of fungal infection should be comprehensively made with clinical, histopathological, and microbiologic evidence. Additionally, new diagnostic modalities including PCR should be considered, as they could provide critical information when the results of other diagnostics are conflicting.