Introduction
Kawasaki disease (KD), which is an acute systemic vasculitis also known as mucocutaneous lymph node syndrome [1,2], is the most common cause of acquired heart disease in children [3]. Potentially serious complications from KD include coronary artery inflammation, aneurysm and possible aneurysm rupture, myocarditis, thrombotic occlusion of the aneurysm, and congestive heart failure [2,3]. According to previous studies, up to 25% of children with KD who go untreated may develop coronary artery abnormalities [4].
Numerous studies have investigated the role of infectious pathogens as potential agents for KD [5,6]. Moreover, studies have attempted to establish a link between KD and specific respiratory viruses including adenovirus, human coronavirus and human bocavirus [7-9]. However, the diagnosis of KD continues to be a challenge and there is still no definitive diagnostic test; accordingly, it is currently being guided based on clinical patient characteristics and the support of laboratory data. Although the etiology of KD remains unknown, some experts believe that infectious exposure leads to the illness [10].
Nevertheless, the role of respiratory viruses in the pathogenesis of KD is still not fully understood [10]. Detection of incidental adenovirus in KD is important to its differentiation from acute adenovirus disease. In a previous study, patients with adenovirus disease were found to have less than 4 KD-like features, predominance of species B or E and higher viral burden compared to those with KD and incidental adenovirus detection [11]. Moreover, Jordan-Villegas et al. [5] reported that human adenoviruses are responsible for 7% to 8% of all pediatric respiratory illness and can lead to prolonged fever, elevated inflammatory markers [11], and clinical features that mimic KD [12]. Acute adenoviral infections have been known to mimic KD, and adenovirus and KD share a number of clinical characteristics; therefore, it can be difficult to distinguish between the two illnesses because the signs and symptoms usually noticed in adenovirus may overlap with those of KD [12,13].
Previous studies have shown that adenovirus might be one of the pathogens that trigger KD [10]. Fukuda et al. [14] reported that simultaneous development of KD followed acute adenovirus infection in monozygotic twins. Moreover, KD can be triggered by adenovirus-3 infection, indicating that specific immune responses to some pathogens such as adenovirus arising from genetic susceptibility may play a critical role in the pathogenesis of KD [14].
Differences in clinical presentations or outcomes have not yet been observed according to positive or negative respiratory viral PCR tests in children with KD [15]. Accordingly, we cannot rule out a diagnosis of KD with a positive respiratory viral PCR or presence of respiratory symptoms at the time of presentation [15]. Because we do not yet have a confirmatory laboratory test that can be used to diagnose KD directly, the diagnostic dilemma is to distinguish adenovirus infection mimicking KD from KD with accompanying adenovirus detection.
Therefore, this retrospective study was conducted to compare the clinical characteristics, laboratory data and echocardiographic findings between simple adenovirus infection and KD with adenovirus infection.
Methods
A retrospective study of 1,403 adenoviral infection patients who were hospitalized at Ewha Womans University Mokdong Hospital from January 2010 to June 2016 was conducted. Only adenoviral infection patients who performed echocardiography was included, and the number of them was 115. And adenoviral infection patients under other respiratory virus coinfection status were also excluded. The enrolled 113 (female 48, male 65) patients were divided into two groups: group 1, which consisted of individuals diagnosed with KD according to the American Heart Association (AHA) criteria (n=62, KD with adenovirus infection); and group 2, which comprised individuals diagnosed only with adenovirus infection (n=51).
This study was approved by the ethics committee of Ewha Womans University Mokdong Hospital Institutional Review Board (2018-03-055-002). The parents of all patients agreed to this study with written informed consent.
1) Diagnosis of adenoviral infection
All patients diagnosed with adenoviral infection were identified by multiplex reverse transcriptase-polymerase chain reaction (RT-PCR), which is a highly sensitive and specific assay. The PCR assay was performed on nasopharyngeal (NP) swabs.
2) Diagnosis of KD
The classical diagnostic criteria for KD as defined by the AHA include the existence of high fever for more than 5 days and the presence of at least 4 of the 5 following criteria: changes in extremities; polymorphous exanthema; bilateral bulbar conjunctival injection without exudates; changes in lips and oral cavity; and cervical lymphadenopathy (>1.5 cm diameter). Incomplete KD can also be diagnosed by the AHA guidelines based on prolonged fever for over 5 days and 2 to 3 of standard clinical features of KD [1].
The following parameters were measured: hemoglobin, white blood cell count, neutrophils percentage, erythrocyte sedimentation rate (ESR), and levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), C-reactive protein (CRP), N-terminal pro-brain natriuretic peptide (NT-proBNP), total protein and albumin. We performed the laboratory examination at the first day of admission to hospital.
The enrolled patients were evaluated by echocardiogram using an IE33 machine (Philips Medical System, Andover, MA, USA) with S8 and S5 transducers during their admission period. Echocardiography approximately performed within 7 to 10 days of onset of fever. Standard parasternal and apical views were acquired. Two dimensional and M-mode echocardiogram, pulsed color-flow Doppler and tissue Doppler imaging (TDI) were obtained. The following left ventricular (LV) parameters were measured by M-mode echocardiography: interventricular septal wall thickness, posterior wall thickness, and LV end diastolic dimension at the chordae tendinae level. The ejection fraction (EF) was determined using the biplane Simpson formula, and fractional shortening (FS) was calculated using LV internal dimensions. The diastolic function was assessed in pulsed Doppler mode from the apical window. Early diastolic velocity (E), late diastolic velocity (A) and E/A ratios were measured using conventional pulsed wave Doppler echocardiography.
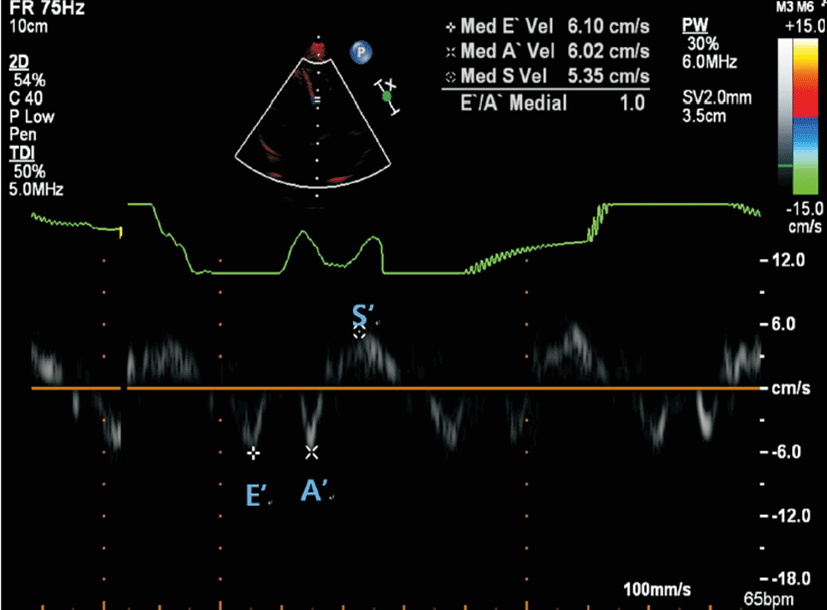
The TDI velocities were obtained at the basal septum from the apical four-chamber view. The Doppler beam was aligned as parallel as possible to the direction of the maximum annular motion. TDI was conducted to obtain longitudinal myocardial velocity with high quality. A narrow sector angle was used, and image depth was adjusted to allow for a high frame rate (130 to 160 frames/sec) with care taken to avoid angulations. The sweep speed was at least 100 mm/sec. The peak systolic myocardial velocity (S’), early diastolic myocardial velocity (E’), and late diastolic myocardial velocity (A’) were obtained (Fig. 1).
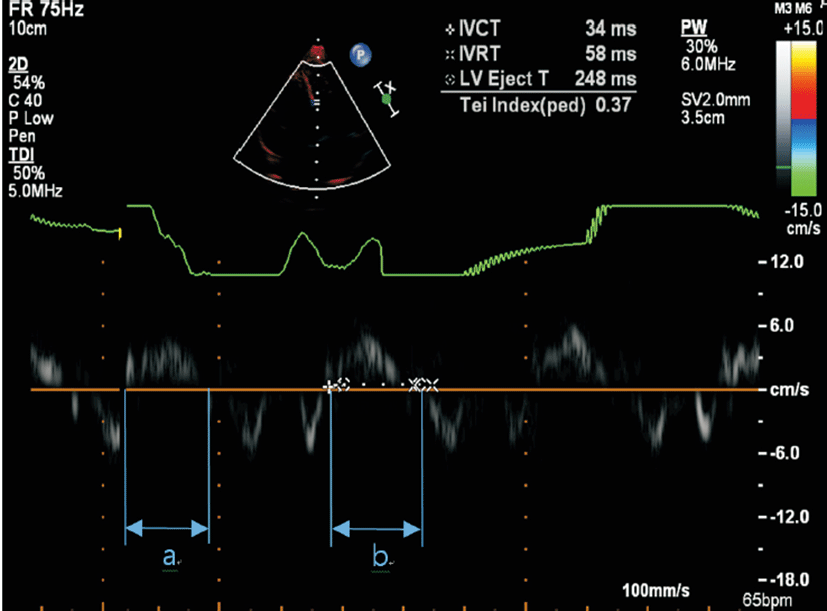
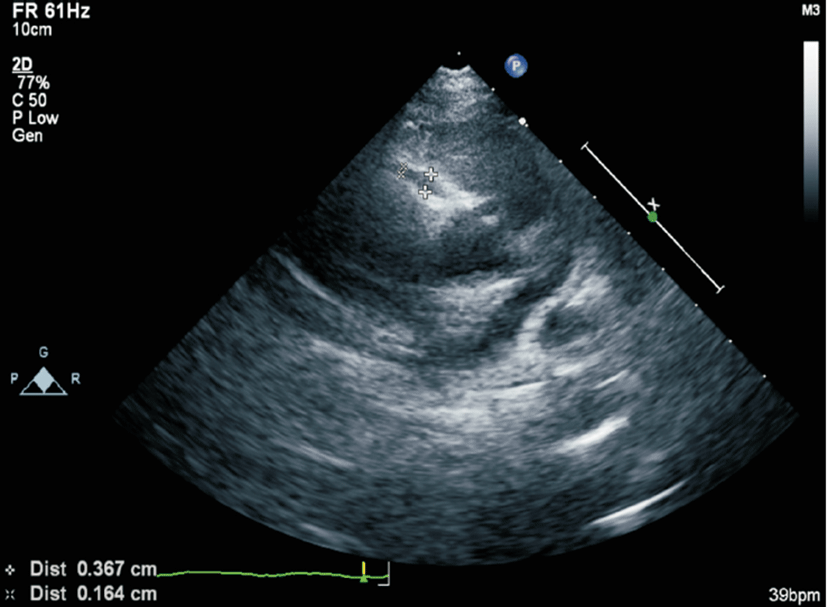
The Tei index, which combines systolic and diastolic LV time intervals, was then calculated from the TDI using the formula (a-b)/b, where a is the interval between the closing and opening of the mitral valve, and b is equal to the left ventricle ejection time (Fig. 1). The isovolumetric relaxation time was measured from the end of the S’ wave to the onset of the E’ wave, and the isovolumetric contraction time was measured from the end of the A’ wave to the onset of the S’ wave (Fig. 2). The coronary artery was measured at the parasternal short axis view (Fig. 3). Coronary arteries were considered abnormal if (1) the internal lumen diameter was >3 mm in children younger than 5 years of age and ≥4 mm in children aged ≥5 years; (2) the internal diameter of a segment was ≥1.5 times that of an adjacent segment; or (3) the coronary lumen was clearly irregular [16].
NP secretions were collected from patients with respiratory symptoms using nasal swabs on the first day of hospitalization. RT-PCR was performed using an AdvanSure RV Real-time PCR kit (LG Life Sciences, Seoul, Korea) with Taqman chemistry technology. This kit consisted of a primer/probe set with sequences specific to each type of viral pathogen. Specifically, the kit targeted 14 strains of RNA virus (corona virus 229E/OC43/NL63, parainfluenza virus 1/2/3, influenza A/B virus, respiratory syncytial virus A/B, rhinovirus A/B/C, and human metapneumovirus) and two strains of DNA virus (adenovirus and bocavirus). The kit enabled sequential RT and multiple PCR within one PCR tube, and was capable of detecting the DNA regions of the bocavirus and the adenovirus. The RNA regions of specific genes of the 14 strains of the RNA virus synthesized complementary DNA through RT, after which complementary DNA created each real-time target PCR product along with specific primers. The Taqman probe fluoresced, and fluorescence intensity was measured using a RT-PCR detection system (SLAN-96P Real-time PCR System; Shanghai Hongshi Medical Technology, Shanghai, China) 0.
PASW Statistics ver. 18.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. Numerical data were expressed as the median (interquartile range) and categorical data were expressed as the number of subjects and percentage. The chi-square test, Fisher exact test and the Mann-Whitney U-test were used to compare between two groups. A P-value <0.05 was considered statistically significant.
Results
In this study, a total of 113 patients (female 48, male 65) were divided into two groups. Group 1 consisted of those who had KD with adenoviral infection and group 2 only had adenoviral infection. Overall, 62 patients (54.8%) were included in group 1 and 51 patients (45.2%) were included in group 2.
The mean patient age was higher in group 1 than group 2 (P=0.045). The sex and fever duration were similar in both groups. However, the following signs were significantly more severe in group 1 than in group 2: conjunctival injection (62.9% vs. 31.4%, P=0.001), cervical lymphadenopathy (32.3% vs. 3.9%, P<0.001), polymorphous skin rash (35.5% vs. 2.0%, P<0.001), abnormalities of lip or oral mucosa (30.6% vs. 3.9%, P<0.001), and abnormalities of extremities (12.9% vs. 2%, P=0.039) (Table 1).
There were no significant differences in hemoglobin, white blood cell count, neutrophils percentage, ESR, serum total protein and NT-proBNP between groups. However, the platelet count (230.0 vs. 257.0 ×109/L, P=0.044), AST (26.5 vs. 32.0 IU/L, P<0.001) and albumin (3.7 vs. 3.8 g/dL, P=0.006) levels were significantly lower in group 1 than group 2. However, the CRP (5.65 vs. 2.52 mg/dL, P<0.001) and ALT (16.5 vs. 15.0 IU/L, P=0.044) levels were significantly higher in group 1 than group 2 (Table 2).
The right coronary artery (RCA) diameter was significantly higher in group 1 than group 2 (2.30 vs. 1.70 mm, P<0.001). The EF and FS were not significantly different between groups. E’ velocity was slightly decreased in group 1 compared with group 2 (9.9 vs. 10.83 cm/sec, P=0.02). There were no significant differences in A’ velocity or S’ velocity. Finally, the Tei index did not differ significantly between two groups (Table 3).
Discussion
Several studies have been conducted to identify the infectious agents responsible for KD [10,17,18]. Respiratory symptoms are frequently observed in children with KD in the acute phase [10]. Turnier et al. [15] reported that 86% of patients with KD had respiratory viral PCR results and 41.9% of them had a positive result in the respiratory viral PCR. The most common virus was rhinovirus/enteroviruses, which was followed by adenovirus, human metapneumovirus and respiratory syncytial virus [15]. Chang et al. [10] suggested that several viruses, including enteroviruses, adenoviruses, rhinoviruses and coronaviruses, were associated with KD. The KD patients in their studies had significantly higher positive rates for virus isolation and PCR than those in the control group (50.4% vs. 16.4%). Jordan-Villegas et al. [5] showed that 8.8% of KD patients had respiratory viruses identified in the upper respiratory tract. Moreover, they reported that patients with KD who had respiratory viruses more frequently showed coronary artery dilatation and were more often diagnosed with incomplete KD. Lee et al. [17] published a study in which the incidence of KD with respiratory symptoms was 31.8%.
The duration of fever was significantly longer and coronary artery diameter was larger in KD with respiratory symptoms compared to KD without respiratory symptoms. Some studies reported an association between specific respiratory viruses and KD [5,18]. However, other authors insisted that the detection of viruses in KD may not have any associative implications [19].
In KD, adenovirus detection by PCR is not uncommon; therefore, quantitative PCR may be helpful in distinguishing adenovirus disease mimicking KD from that with concomitant adenovirus detection [18,20]. Primary infections with adenovirus C species commonly occur in children younger than age 5. After initial infection, adenovirus C can also persist in the tonsils for several years with low grade replication [9]. Jaggi et al. [18] suggested that detection of adenovirus in a patient with suspected KD should be interpreted cautiously because detection of adenovirus is not uncommon; therefore, a diagnosis of KD should not be excluded. PCR is an important diagnostic tool because it is highly sensitive and can detect low-level viruses [18].
Other clinical or laboratory data that are useful in distinguishing KD from adenoviral disease include neck swelling, distal extremity changes and presence of pyuria and/or hepatitis [18]. A positive result for a respiratory virus, regardless of the virus detected, should not be used to exclude the diagnosis of KD [18]. Continued research is needed to elucidate the etiology and discover a more sensitive and specific diagnostic test for this important KD [15].
Human adenoviruses are detected in 7% to 8% of all pediatric respiratory illnesses [9]. Adenovirus species C is a cause of pediatric illness. Because it is commonly detected in the nasopharynx incidentally because of persistence in the tonsil and adenoid tissue, its presence can lead to misinterpretation of a positive result in an acute illness [21]. Additionally, PCR has replaced adenoviral culture as a diagnosis, which has also led to confusion in diagnosis between adenoviral infection and KD because of the highly sensitive PCR test. This incidental detection by PCR of adenovirus has been observed in patients with KD, some of whom developed coronary artery abnormalities [5].
Adenoviruses have been reported in the NP secretions of 2% to 9% of patients with KD [5,10,18]. Song et al. [11] reported that 8.8% of patients with complete criteria for diagnosis of KD were also found to have adenovirus in the respiratory tract upon PCR analysis. Furthermore, they found that their patients with complete KD had lower viral burden and a higher percentage of species C than those with adenoviral infection. Adenoviruses have been detected by PCR in the NP secretions of up to 11% of healthy children;[11] therefore, detection of adenovirus by PCR in the NP secretion does not mean infection.
Some patients developed coronary artery aneurysm [5], clearly indicating that detection of adenovirus in the respiratory tract does not exclude the diagnosis of KD. However, adenovirus illness can also mimic KD with prolonged fever, other similar clinical features and elevated inflammatory markers [22,23]. Ideal diagnostic tools to make a completely accurate diagnosis may not be available, but we found certain clinical, laboratory, and virologic characteristics that aid in differentiation of these two conditions.
Children with acute adenovirus disease are very likely to have less than four KD-like clinical features. Adenovirus conjunctivitis includes unilateral onset, prominence of tearing more frequently than purulence and follicular hyperplasia [11]. However, as noted in previous studies, the least common finding was changes in the extremities [1,24]. In our study, the adenoviral infection group most commonly had fever (100%) and conjunctival injection (31.4%). However, cervical lymphadenopathy (3.9%), abnormality of lip or oral mucosa (3.9%) and rash (2%) were rare in adenoviral infection compared with the KD group. The signs were more severe in KD compared than only adenoviral infection such as conjunctival injection (62.9% vs. 31.4%, P=0.001), cervical lymphadenopathy (32.3% vs. 3.9%, P=0.000), polymorphous skin rash (35.5% vs. 2.0%, P=0.000), abnormalities of lip or oral mucosa (30.6% vs. 3.9%, P=0.000) and abnormalities of extremities (12.9% vs. 2%, P=0.039).
The AHA algorithm is very useful in diagnosing incomplete KD. However, the current guidelines suggest that a finding of CRP of ≥3 mg/dL or ESR ≥40 mm/hr [1,2] indicates a diagnosis of incomplete KD. Nevertheless, several studies have documented higher levels of these inflammatory markers in adenovirus patients [22-24]. In the present study, the mean CRP level was 3.35 mg/dL in the adenoviral group. We believe this suggests that this CRP level may be inadequate for discriminating between the two diseases. These findings are consistent with those of Song et al. [11], who reported that CRP was consistently higher in subjects with KD compared with those who had adenovirus, but that both had a median CRP of ≥3 mg/dL, suggesting this threshold for discrimination may lack specificity for KD. In our study, CRP, ALT and RCA diameter were significantly higher in group 1 than group 2. Although the level of ALT don not have clinical difference on account of within the normal range in both groups, the other significant findings, CRP and dilated RCA, can be helpful to differential diagnosis. In addition, making a correct diagnosis is necessary because untreated cases may lead to coronary artery damage. Because adenoviral infection has similar clinical symptoms, laboratory findings and echocardiography are needed to differentiate between these two diseases [25].
It should be noted that there are some limitations to this study. Specifically, the sample size was relatively small. PCR was only performed on KD patients with respiratory symptoms because of the relative expense of the PCR test. For a complete analysis, PCR should have been performed on all patients; however, not all parents of the patients agreed to the test. Moreover, this study was a retrospective analysis. Accordingly, further prospective studies are needed to clarify the differential diagnosis between adenoviral infection and KD with incidental adenoviral detection.
In conclusion, in patients with adenoviral infection with unexplained prolonged fever, if patients represent typical KD features, we can diagnosis by clinical manifestations. Otherwise we need to consider another way to make a correct diagnosis. We should consider performing echocardiogram to find out whether coronary arteries are dilated. The dilated RCA or high level of CRP can be used to differentiate KD with adenoviral infection from adenoviral infection alone.