Abstract
A 28-year-old woman developed drug-induced tachycardia in the left lateral decubitus (LLD) position following intravenous administration of glycopyrrolate. The tachycardia was unresponsive to esmolol, labetalol, and fentanyl but resolved unexpectedly after the tidal volume was reduced from 8 to 6 mL/kg. Drug-induced tachycardia or arrhythmia occurring in the LLD position under mechanical ventilation may be attenuated by adopting a low tidal volume ventilation strategy (6 mL/kg).
-
Keywords: Heart rate; Cholinergic antagonists; Ventilation; Anesthesia; Posture; Autonomic nervous system; Case reports
Introduction
Background
The cardiovascular system regulates heart rate (HR) and arterial blood pressure via the autonomic nervous system (ANS) in response to physiological and environmental changes. Some studies have reported that a recumbent position may influence ANS activity and hemodynamics in patients with congestive heart failure (CHF) [
1,
2] and might also affect healthy individuals [
3].
Glycopyrrolate, a muscarinic anticholinergic commonly used preoperatively to reduce respiratory secretions, may cause systemic side effects such as mydriasis, tachycardia, prostration, anorexia, and diarrhea [
4].
This case suggests that even in patients without a cardiovascular history, administration of anticholinergic agents in the left lateral decubitus (LLD) position could increase cardiac burden, potentially resulting in arrhythmia or drug-resistant tachycardia. Such complications may be mitigated by reducing the tidal volume to approximately 6 mL/kg.
Objectives
We report a case of a 28-year-old woman who developed glycopyrrolate-induced sinus tachycardia in the LLD position that was unresponsive to esmolol, labetalol, and fentanyl, but unexpectedly resolved shortly after tidal volume reduction.
Case presentation
Ethics statement
This study was approved by the Institutional Review Board (IRB) of the Korea Institute of Radiological & Medical Sciences (IRB No. 2025-10-011). The IRB approved a waiver of informed consent.
A 28-year-old woman (156.2 cm, 50.8 kg) was scheduled for curettage and bone cementing for a giant cell tumor of the right distal femur (6.7×7.3×5.9 cm). Preoperative laboratory tests revealed mild hyponatremia (126 mmol/L) and hypoglycemia (65 mg/dL), which were corrected the day before surgery (sodium: 133 mmol/L, glucose >70 mg/dL). Her past medical history, pulmonary function test results, and preoperative electrocardiogram were unremarkable.
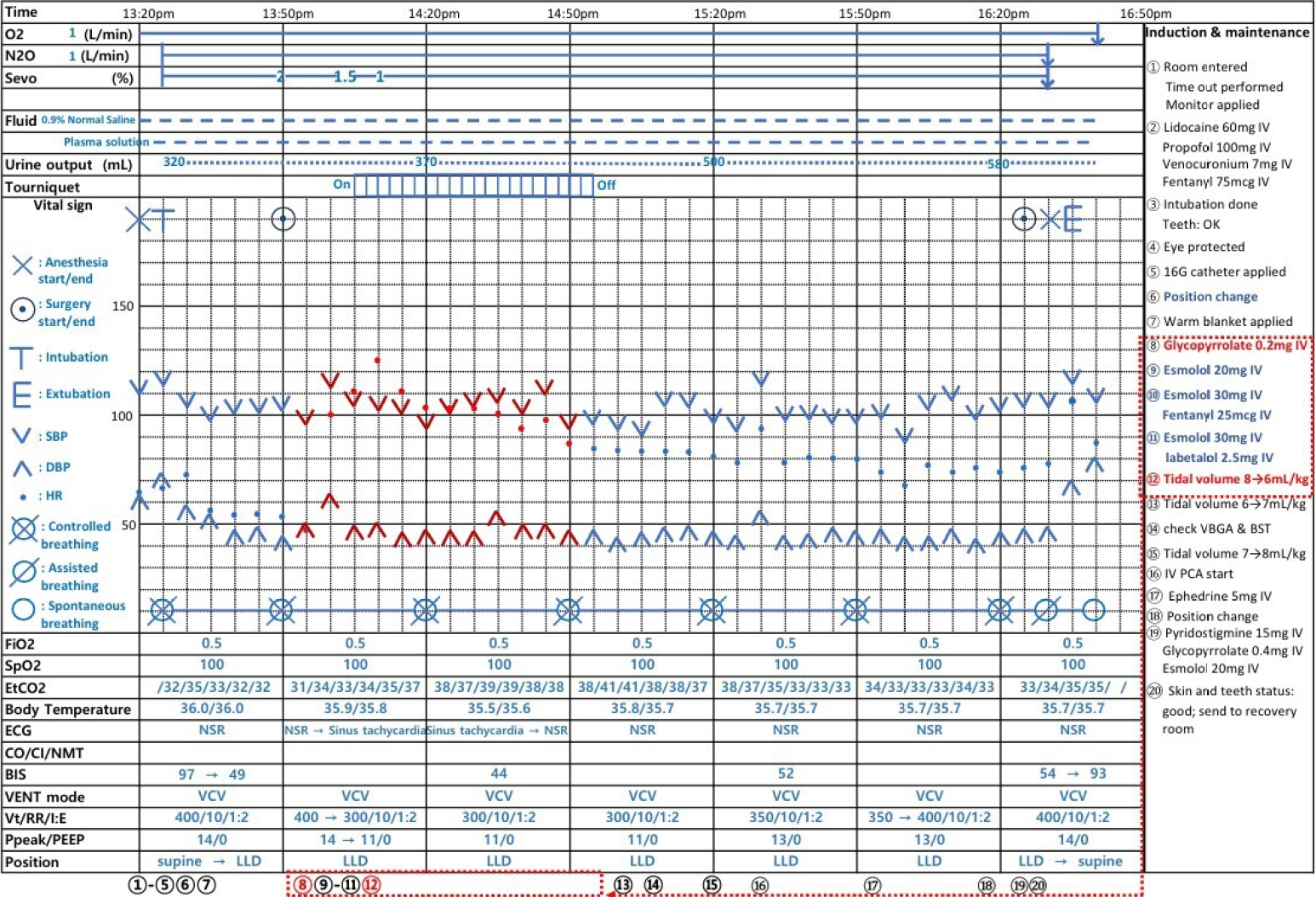
On the day of surgery, no premedication was administered. Upon arrival in the operating room, the patient’s peripheral oxygen saturation (SpO2) was 99%, blood pressure was 110/62 mm Hg, and HR was 64 beats per minute (bpm). Preoxygenation was performed using face mask ventilation with 100% oxygen at a flow rate of 6 L/min. General anesthesia was induced with lidocaine 50 mg, propofol 100 mg, vecuronium 7 mg, and fentanyl 75 µg. Tracheal intubation was performed using a 7.0-mm endotracheal tube. After intubation, bilateral breath sounds were confirmed to be clear without abnormalities, and mechanical ventilation was initiated in volume-controlled ventilation mode. Anesthesia was maintained with sevoflurane 1.5% and nitrous oxide 50% in oxygen at a total flow rate of 2 L/min. Tidal volume was set at 8 mL/kg with a respiratory rate of 10 breaths/min. Ventilation and oxygenation were monitored using end-tidal CO2 (EtCO2) and SpO2. Blood pressure was measured with a non-invasive blood pressure cuff on the left arm. A peripheral intravenous line was secured in the right arm via a 16-gauge catheter.
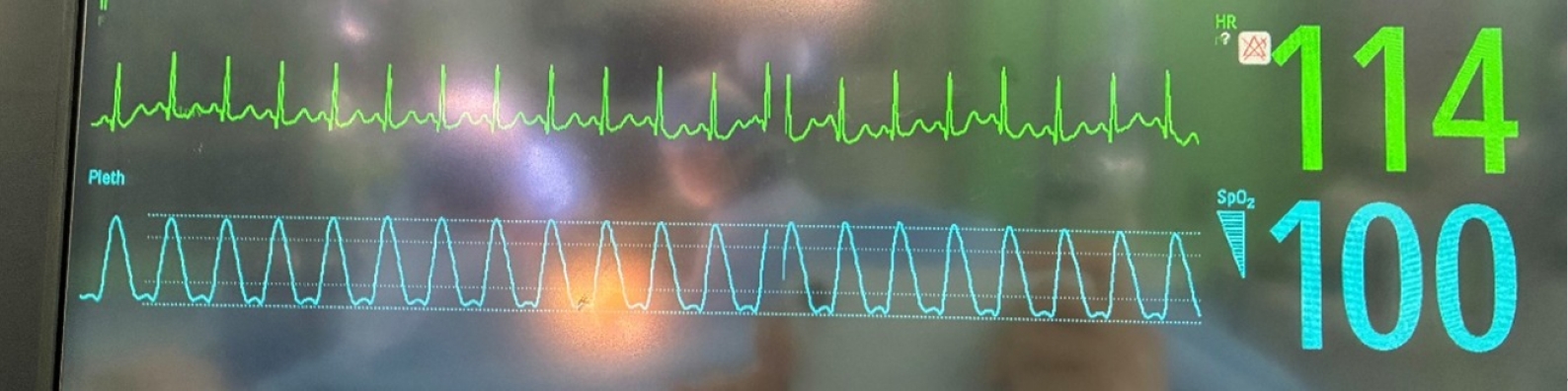
The patient was placed in the LLD position, and a tourniquet was applied to the right thigh. She was positioned near the left edge of the operating table. The dependent leg was well-padded, and an additional pad was placed between the legs for stability. The patient was then secured with a surgical table strap to prevent unintended movement. From that point onward, the mean arterial pressure (MAP) remained at approximately 66 mm Hg, while HR persisted at 49–55 bpm. Therefore, glycopyrrolate 0.2 mg was administered intravenously to increase the HR, as glycopyrrolate, compared with atropine, typically produces smaller increases in HR and the rate–pressure product, a hemodynamic parameter considered a reliable surrogate for myocardial oxygen demand [
5]. HR increased to over 100 bpm immediately after glycopyrrolate administration (
Fig. 1); thus, esmolol 20 mg was administered. However, HR continued to rise, requiring repeated doses of esmolol (30 mg ×2) without effect (
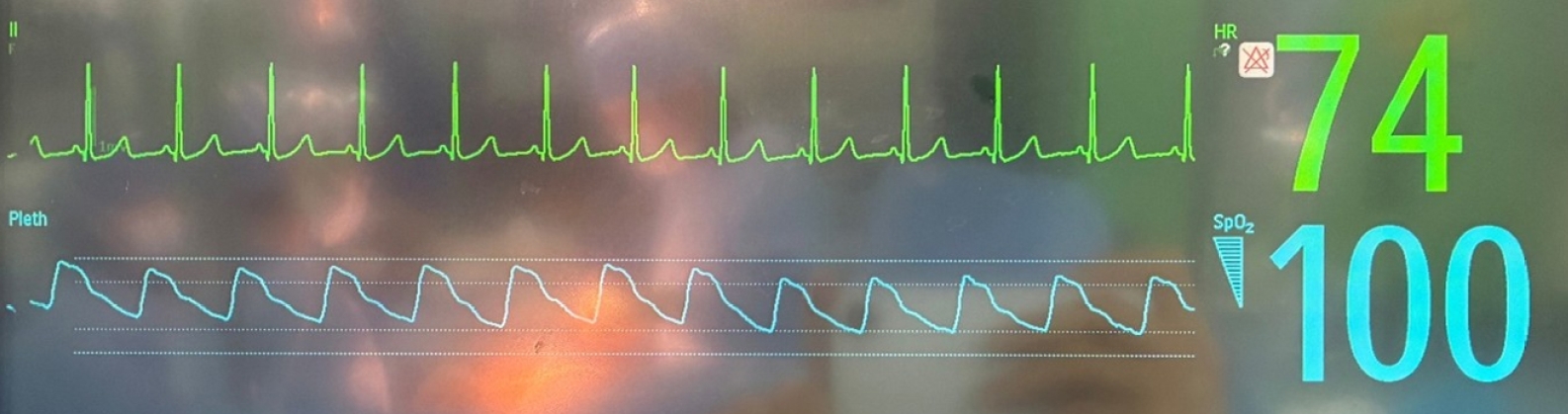
Fig. 2). HR subsequently peaked at 122 bpm despite additional administration of labetalol 2.5 mg and fentanyl 25 µg, both given to attenuate the tachycardic response. During this period, systolic blood pressure was maintained at 101–110 mm Hg, diastolic blood pressure at 50–62 mm Hg, and MAP at 65–75 mm Hg. Sevoflurane was reduced to 1%, and the team notified the attending anesthesiologist, who recommended increasing tidal volume to 9–10 mL/kg to prevent hypercarbia. However, due to miscommunication, tidal volume was instead reduced to 6 mL/kg while sevoflurane was maintained at 1%; during this time, EtCO
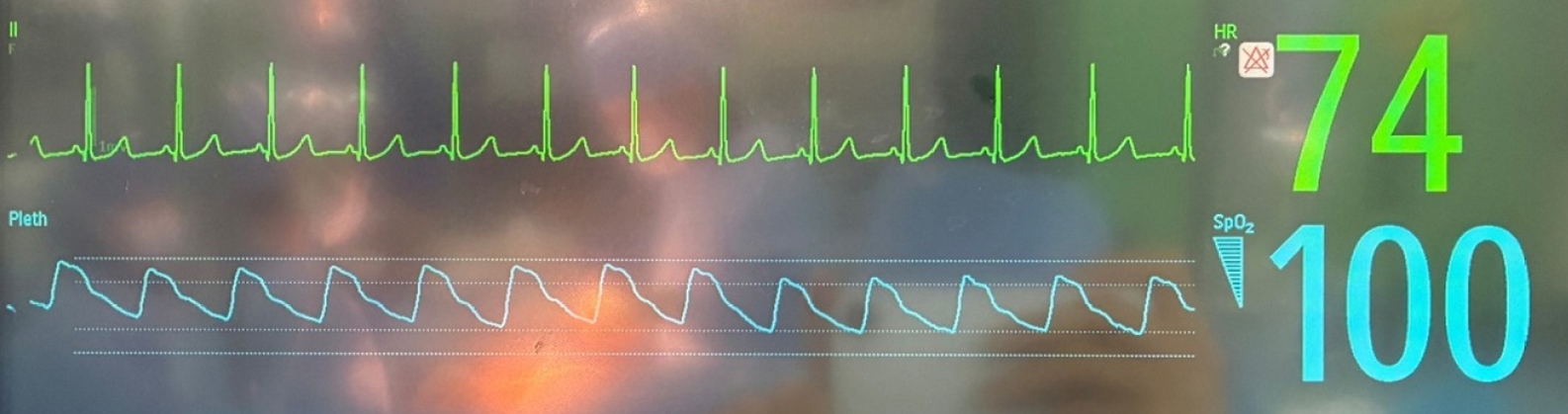
2 remained at 39–41 mm Hg. Remarkably, HR promptly decreased to 109 bpm and returned below 100 bpm within 25 minutes (
Fig. 3), while MAP remained near 66 mm Hg. Glycopyrrolate-induced tachycardia persisted for approximately 40 minutes. Thereafter, tidal volume was adjusted to 7–8 mL/kg to maintain a target EtCO
2 of approximately 35 mm Hg.
During emergence, the patient was repositioned supine, and neuromuscular blockade was reversed with glycopyrrolate 0.4 mg and pyridostigmine 15 mg. She was extubated uneventfully, with no further complications.
Discussion
In our case, after the patient was placed in the LLD position, HR initially decreased to 49–53 bpm. Chen and Kuo [
3] reported that cardiac vagal tone varies by recumbent body position, based on spectral heart rate variability (HRV) indices such as normalized high-frequency power and the low-/high-frequency (LF/HF) power ratio, suggesting that autonomic modulation may differ with posture even in healthy individuals. Miyamoto et al. [
1] described no significant differences in the LF/HF ratio among 3 recumbent positions (supine, LLD, and RLD) in healthy participants, indicating that cardiac autonomic activity is unaffected by posture, unlike in patients with CHF; however, Sasaki et al. [
6] demonstrated that HR reduction and an increase in left ventricular end-diastolic diameter in the LLD position, without significant changes in stroke volume, cardiac output, or HRV, represent a physiological phenomenon rather than altered ANS activity.
Several mechanisms for positional effects on cardiac function have been proposed. First, gravitational effects in the LLD position may displace the heart, leading to distortion of the pulmonary veins and impaired venous return. Second, in patients with cardiomegaly, the LLD position may cause left ventricular compression against the chest wall, impairing diastolic filling. A recent case report by Feng et al. [
7] further described this phenomenon, reporting severe hypotension and trepopnea in the LLD position during preparation for spinal anesthesia. These findings suggest that the reduction in HR in the LLD position may relate to increased cardiac burden.
When HR increased after glycopyrrolate administration, the resulting sinus tachycardia was refractory to pharmacologic interventions (e.g., esmolol, labetalol, fentanyl) but resolved after tidal volume was reduced from 8 to 6 mL/kg. De Backer et al. [
8] identified tidal volume as a major determinant of right ventricular (RV) afterload and stroke work, while Mahmood and Pinsky [
9] demonstrated that large tidal volumes increase intrathoracic pressure swings, impairing RV ejection due to limited contractile reserve. In this case, an intermediate tidal volume (8 mL/kg), combined with positional effects, may have compromised RV function and facilitated glycopyrrolate-induced tachycardia. Cherpanath et al. [
10] further reported that intermediate tidal volumes (8–10 mL/kg) cause biventricular systolic dysfunction compared with a low tidal volume (4–6 mL/kg) strategy, likely due to ventilation-induced inflammation, without affecting diastolic function. Thus, reducing the tidal volume to 6 mL/kg likely improved cardiac performance and contributed to HR normalization in this case.
In addition, Natalini et al. [
11] reported that a low tidal volume ventilation strategy (6 mL/kg of ideal body weight) improved cardiac index and oxygen delivery compared with a high tidal volume strategy (12 mL/kg of ideal body weight) in patients with acute respiratory distress syndrome. These effects were attributed to a slight increase in arterial carbon dioxide tension (PaCO
2), which reduces systemic vascular resistance and enhances pulmonary vasoconstriction, thereby improving ventilation–perfusion matching [
12].
This analysis is limited by the absence of invasive hemodynamic monitoring (e.g., an arterial line, central venous line, or Swan–Ganz catheter), as the procedure was not extensive and the patient had no significant past medical history. Nonetheless, intraoperative hypovolemia was considered unlikely because urine output remained above 0.5 mL/kg/hr, and venous blood gas analysis performed approximately 1 hour after the onset of tachycardia showed a hemoglobin level of 9.9 g/dL. As other potential causes were evaluated, attention was directed toward the possibility of a drug-induced reaction. Choi and Kim [
13] reported an anaphylactic reaction following the administration of glycopyrrolate and pyridostigmine, which presented as refractory hypotension and an erythematous skin rash. In contrast, in our case, an adverse drug reaction such as glycopyrrolate-induced anaphylaxis was excluded, as no abnormal findings were observed either during emergence following administration of the reversal agent or after subsequent administration of glycopyrrolate for bradycardia. Consequently, the resolution of drug-induced tachycardia after tidal volume reduction may suggest that modulation of intrathoracic pressure improved cardiac performance, consistent with previous reports in the literature.
In conclusion, drug-induced tachycardia or arrhythmia occurring in the LLD position under mechanical ventilation may be attenuated by low tidal volume ventilation (6 mL/kg).
-
Authors’ contribution
Conceptualization: SJA. Data curation: SJA. Methodology/formal analysis/validation: SJA, SYL. Project administration: SJA. Funding acquisition: SJA. Writing–original draft: SJA. Writing–review & editing: SJA, SYL.
-
Conflict of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Not applicable.
-
Acknowledgments
None.
Fig. 1.Timetable of anesthetic care during the operation. BIS, bispectral index; BP, blood pressure; SBP, systolic blood pressure; DBP, diastolic blood pressure; ECG, electrocardiogram; HR, heart rate; IV-PCA, intravenous patient-controlled analgesia; NMT, neuromuscular transmission; NSR, normal sinus rhythm; Ppeak/PEEP, peak inspired airway pressure/positive end expiratory pressure; RR, respiration rate; VENT mode, ventilation; VCV, volume-controlled ventilation; Vt, tidal volume; EtCO2, end-tidal CO2; FiO2, fraction of inspired oxygen; SpO2, peripheral oxygen saturation.
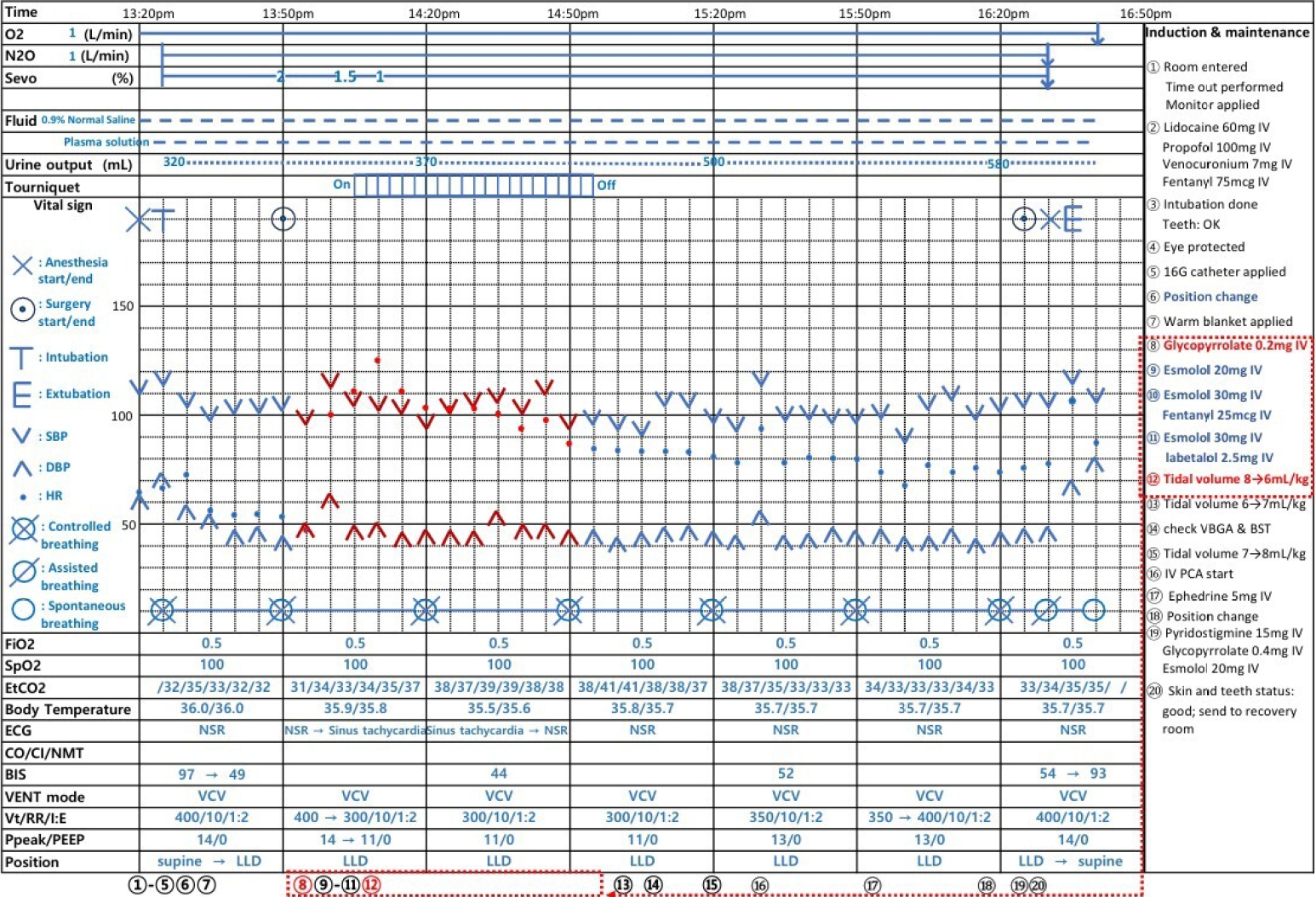
Fig. 2.Electrocardiogram demonstrating increased heart rate immediately after glycopyrrolate administration.
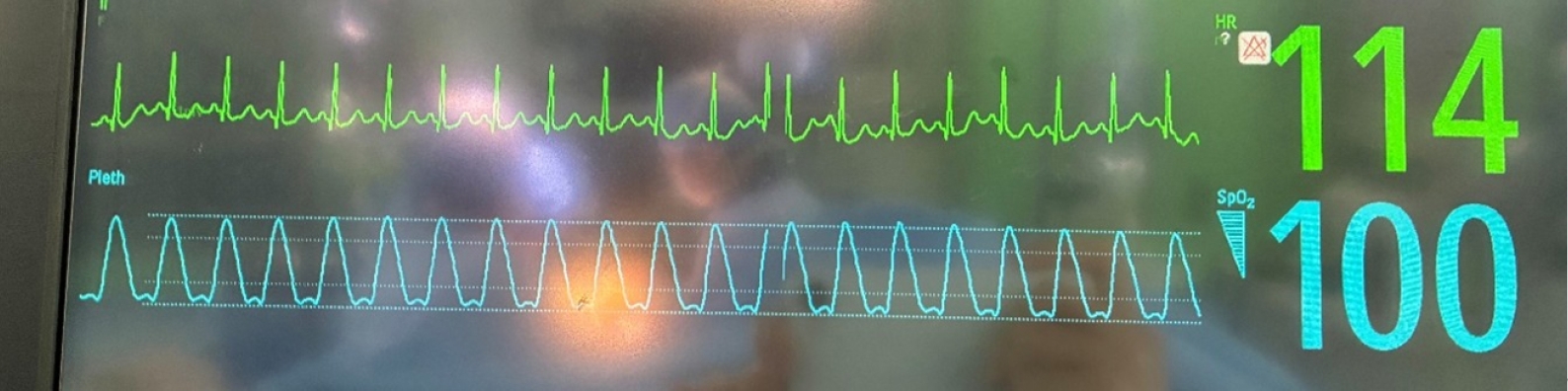
Fig. 3.Electrocardiogram demonstrating heart rate 1 hour after tidal volume reduction.
References
- 1. Miyamoto S, Fujita M, Sekiguchi H, Okano Y, Nagaya N, Ueda K, Tamaki S, Nohara R, Eiho S, Sasayama S. Effects of posture on cardiac autonomic nervous activity in patients with congestive heart failure. J Am Coll Cardiol 2001;37:1788-1793. https://doi.org/10.1016/s0735-1097(01)01249-9
- 2. Fujita M, Miyamoto S, Sekiguchi H, Eiho S, Sasayama S. Effects of posture on sympathetic nervous modulation in patients with chronic heart failure. Lancet 2000;356:1822-1823. https://doi.org/10.1016/S0140-6736(00)03240-2
- 3. Chen GY, Kuo CD. The effect of the lateral decubitus position on vagal tone. Anaesthesia 1997;52:653-657. https://doi.org/10.1111/j.1365-2044.1997.114-az0106.x
- 4. Chabicovsky M, Winkler S, Soeberdt M, Kilic A, Masur C, Abels C. Pharmacology, toxicology and clinical safety of glycopyrrolate. Toxicol Appl Pharmacol 2019;370:154-169. https://doi.org/10.1016/j.taap.2019.03.016
- 5. Mostafa SM, Vucevic M. Comparison of atropine and glycopyrronium in patients with pre-existing cardiac disease. Anaesthesia 1984;39:1207-1213. https://doi.org/10.1111/j.1365-2044.1984.tb06433.x
- 6. Sasaki K, Haga M, Endo Y, Fujiwara J, Maruyama R. Left recumbent position decreases heart rate without alterations in cardiac autonomic nervous system activity in healthy young adults. Tohoku J Exp Med 2017;241:309-318. https://doi.org/10.1620/tjem.241.309
- 7. Feng CW, Liu YT, Lin TC. Deteriorating hypotension and trepopnea during left lateral decubitus position before spinal anesthesia. Asian J Anesthesiol 2024;62:95-97. https://doi.org/10.6859/aja.202406_62(2).0004
- 8. De Backer D, Heenen S, Piagnerelli M, Koch M, Vincent JL. Pulse pressure variations to predict fluid responsiveness: influence of tidal volume. Intensive Care Med 2005;31:517-523. https://doi.org/10.1007/s00134-005-2586-4
- 9. Mahmood SS, Pinsky MR. Heart-lung interactions during mechanical ventilation: the basics. Ann Transl Med 2018;6:349. https://doi.org/10.21037/atm.2018.04.29
- 10. Cherpanath TG, Simonis FD, Bouma BJ, de Bruin-Bon RH, Determann RM, Juffermans NP, Gama de Abreu M, Pelosi P, Serpa Neto A, Groeneveld JA, Schultz MJ, Lagrand WK. Myocardial function during low versus intermediate tidal volume ventilation in patients without acute respiratory distress syndrome. Anesthesiology 2020;132:1102-1113. https://doi.org/10.1097/ALN.0000000000003175
- 11. Natalini G, Minelli C, Rosano A, Ferretti P, Militano CR, De Feo C, Bernardini A. Cardiac index and oxygen delivery during low and high tidal volume ventilation strategies in patients with acute respiratory distress syndrome: a crossover randomized clinical trial. Crit Care 2013;17:R146. https://doi.org/10.1186/cc12825
- 12. Kregenow DA, Swenson ER. The lung and carbon dioxide: implications for permissive and therapeutic hypercapnia. Eur Respir J 2002;20:6-11. https://doi.org/10.1183/09031936.02.00400802
- 13. Choi E, Kim S. Anaphylactic reaction following reversal of nondepolarizing muscle relaxant during general anesthesia: a case report. Int Med Case Rep J 2017;10:271-274. https://doi.org/10.2147/IMCRJ.S142597