Abstract
Itching is a representative manifestation of urticaria. However, under certain conditions, urticaria may be characterized by nociceptive sensations, such as stinging or tingling, either instead of or in addition to itching. Three patients with chronic urticaria (CU) who experienced nociceptive pain accompanied by itching were identified and treated with immunoglobulin/histamine complex (IHC) therapy. Nociceptive pain resolved along with improvement in CU symptoms after 8 injections of IHC in the first patient and after 4 injections in the second and third patients. Nociceptive pain may present as a symptom of CU. The clinical characteristics of the transition from itching to nociceptive pain, together with the observed outcomes of IHC therapy, appear to support the intensity theory explaining this shift, which may be mediated by histamine. Further clinical and basic immunological studies are warranted to clarify the underlying mechanisms.
-
Keywords: Chronic urticaria; Histamine; Immunoglobulin; Nociceptive pain; Case reports
Introduction
Itching is a characteristic and typical manifestation of urticaria, including chronic urticaria (CU) [
1]. However, under certain conditions, urticaria may present with nociceptive pain, such as stinging or tingling, either instead of or in addition to itching [
2].
Histamine plays a central role in the pathogenesis of CU [
3]. In addition, histamine is a known mediator of pain sensation [
4]. Both itching and nociceptive signals are typically transmitted through C-fibers [
5].
Histobulin (Green Cross PD) is an immunoglobulin/histamine complex (IHC) preparation comprising 0.15 µg of histamine dihydrochloride and 12 mg of immunoglobulin G [
6]. It was developed to regulate serum histamine levels through histaminopexy [
7]. IHC is effective in the treatment of allergic rhinitis, bronchial asthma, CU, and atopic dermatitis [
8].
IHC therapy has been reported to be effective for the treatment of CU [
9,
10]. Its therapeutic effects are progressive and gradual, in contrast to those of other treatments, including omalizumab and antihistamines, which typically produce more rapid symptom relief [
11,
12]. Recent studies have suggested that IHC may induce remission and have proposed IHC therapy as a potentially effective therapeutic modality for CU [
8].
The primary mechanism of action of IHC is histaminopexy, which involves regulation of circulating histamine levels through inhibition of histamine release from mast cells and induction of antibodies against histamine [
13-
15].
Given current understanding of the pathophysiology of histamine-mediated nociceptive signaling [
4,
5], the clinical presentations of CU—itching and nociceptive pain—may represent closely related manifestations rather than entirely distinct symptoms.
This report describes 3 patients with CU who presented with nociceptive pain. The clinical characteristics of itching and nociceptive pain in these patients, as well as their changes during IHC therapy, are detailed.
Case presentation
Ethics statement
Informed consent was obtained from all patients.
Case 1
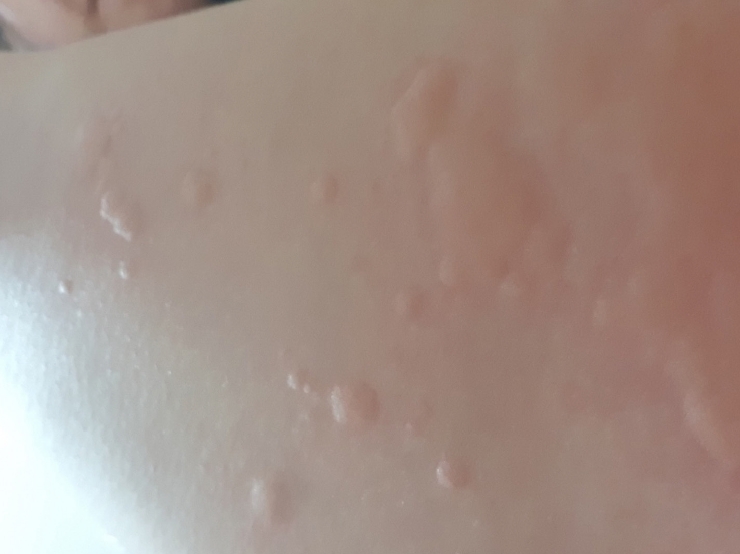
The first patient was a 39-year-old Korean woman who presented to the Department of Allergy and Clinical Immunology at Cheju Halla General Hospital with urticaria and itching accompanied by a tingling sensation described as nociceptive pain for 1 month (
Fig. 1). She reported recurrent episodes of nociceptive pain associated with itching. There was no personal or family history of CU.
Because the patient’s symptoms persisted for more than 6 weeks during evaluation, a diagnosis of CU was established according to accepted diagnostic criteria [
1]. Following this diagnosis, laboratory evaluations were performed, including a complete blood count with differential and measurements of serum eosinophil cationic protein, total serum immunoglobulin E (IgE), and allergen-specific IgE using a multiple allergosorbent test (MAST; Green Cross PD). Serum immunoglobulin A levels were also measured prior to initiation of IHC therapy (
Table 1). In the MAST, specific IgE levels for 41 allergens were assessed, and values <0.35 IU/mL were considered negative. A skin prick test was also performed for 53 allergens. Histamine hydrochloride (10 mg/mL) served as the positive control, and normal saline served as the negative control. Wheal diameters were measured, and reactions were read after 15 minutes. Responses were graded as follows: negative (0, no reaction); 1+ (reaction greater than the control but less than half the size of the histamine reaction); 2+ (equal to or more than half the size of the histamine reaction); 3+ (equal to or more than the size of the histamine reaction); or 4+ (equal to or more than twice the size of the histamine reaction). A wheal size of ≥3 mm was considered positive. The patient was instructed to discontinue antihistamines for at least 5 days before testing.
All allergy laboratory tests yielded results within normal ranges, including the eosinophil fraction on complete blood count, serum eosinophil cationic protein, and total serum IgE levels (
Table 1). No allergen-specific IgE levels were elevated on MAST, and no positive reactions were observed on skin prick testing.
The clinical severity of CU was assessed using the Urticaria Severity Scale described by Jariwala et al. [
16], which has a maximum possible score of 93 points. Pain severity was evaluated using a numerical rating scale with a maximum score of 10 [
2].
Histobulin (Green Cross) was used as the IHC preparation. Each 2-mL ampule contains 12 mg of human immunoglobulin and 0.15 µg of histamine [
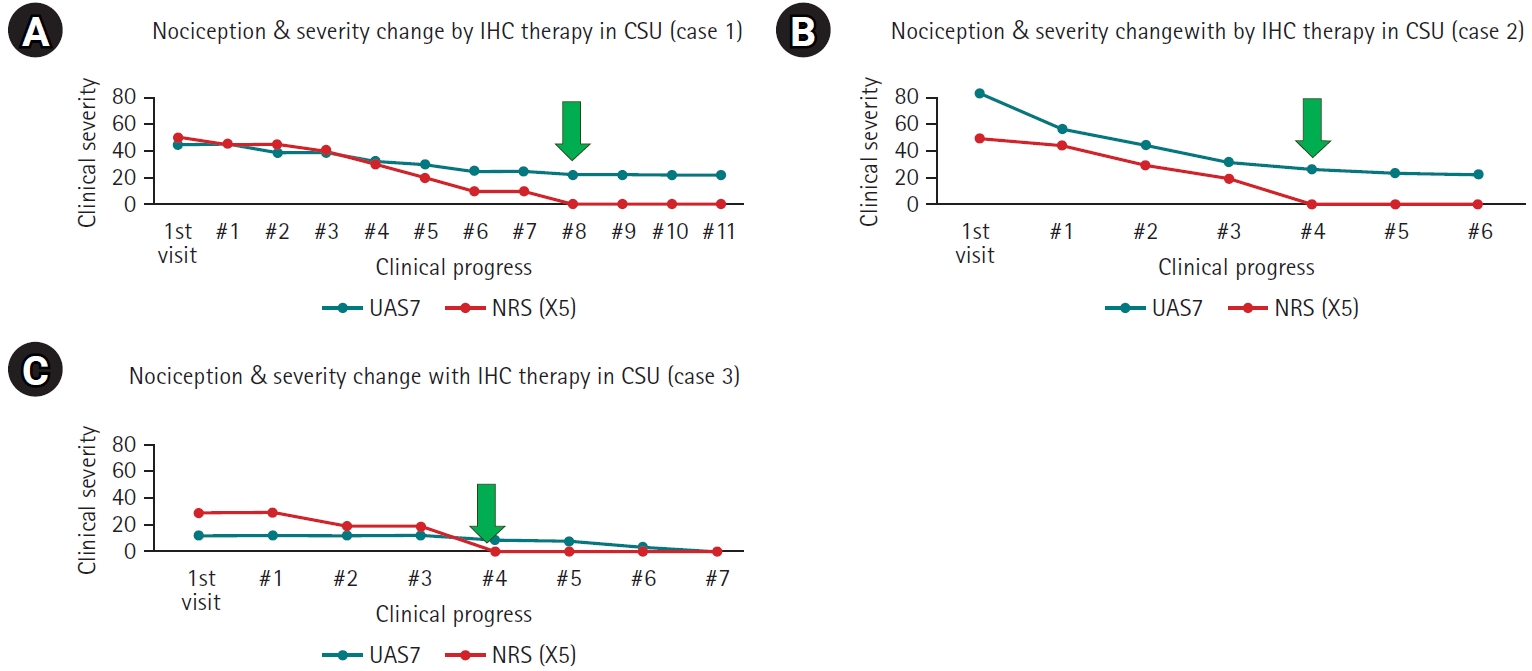
6]. IHC was administered by weekly subcutaneous injection into the deltoid region of the upper arm until remission was achieved. During therapy, the patient was instructed to take levocetirizine 5 mg once daily as needed if symptoms interfered with activities of daily living, work, or sleep. Nociceptive pain resolved as CU symptoms improved following 8 IHC injections (
Fig. 2A).
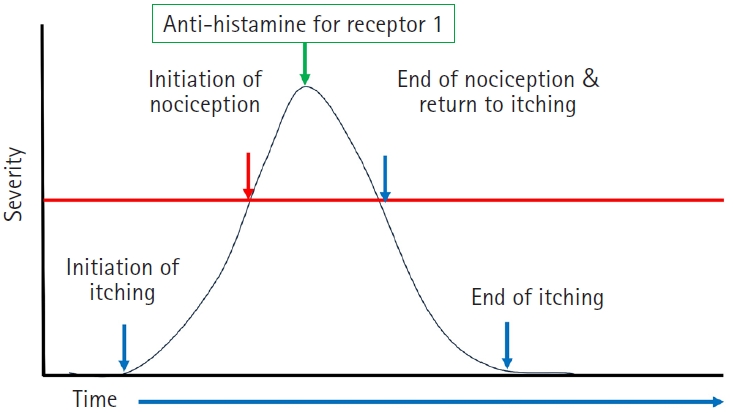
During symptomatic episodes, itching developed first and was followed shortly thereafter by nociceptive pain (
Fig. 3). In all 3 patients, administration of levocetirizine led to resolution of nociceptive pain; itching subsequently reappeared briefly before gradually subsiding during each episode.
The second patient was a 42-year-old Korean woman who presented to the Department of Allergy and Clinical Immunology at Cheju Halla General Hospital with urticaria, combined itching and stinging described as a nociceptive sensation, and frequent angioedema of the lips for 4 months. She had no personal history of CU; however, her brother had previously experienced urticaria and itching. She underwent the same basic allergy evaluation as described in case 1, including MAST and skin prick testing.
No abnormal laboratory findings were identified except for elevated allergen-specific IgE levels to
Dermatophagoides pteronyssinus,
Dermatophagoides farinae, and cockroach on MAST and positive skin reactivity on skin prick testing (
Table 1). IHC therapy was initiated, and nociceptive pain resolved as CU symptoms improved after 4 IHC injections (
Fig. 2B).
The third patient was a 57-year-old Japanese woman who presented to the Department of Allergy and Clinical Immunology at Cheju Halla General Hospital with urticaria and combined itching and a tingling sensation described as a nociceptive symptom. Her history included several prior episodes of urticaria. She also reported a family history of urticaria in both parents, and her grandmother had atopic dermatitis. Basic allergy testing with MAST and skin prick tests was performed as described in case 1.
Laboratory testing showed allergen-specific IgE to multiple allergens, including
D. pteronyssinus,
D. farinae, and pollens, on MAST, along with positive skin reactivity on skin prick testing (
Table 1). IHC therapy was administered, and CU symptoms improved. After 4 IHC injections, the patient’s nociceptive pain resolved as her CU presentation improved (
Fig. 2C).
Discussion
As shown in this report, some patients with CU may experience nociceptive pain in addition to itching. In all 3 patients, nociceptive pain resolved as CU symptoms improved following IHC therapy (
Fig. 2).
The primary mechanism of action of IHC is thought to involve histaminopexy, including histamine-fixing effects that promote the production of antihistamine antibodies [
13]. Nociceptive pain has been reported as a symptom of cholinergic urticaria [
2]. Histamine plays a central role in the pathogenesis of CU [
3] and is also a known mediator of pain sensation [
4]. Accordingly, some patients with CU may experience nociceptive symptoms, including pain, either instead of or in addition to itching. In these cases, the resolution of nociceptive pain following IHC therapy suggests that histamine may contribute to nociceptive pain in CU (
Fig. 2). Levocetirizine also improved both itching and nociceptive pain (
Fig. 3), suggesting involvement of histamine receptor 1. Histamine receptor 1 activation increases intracellular Ca2+, which activates protein kinase C–dependent phosphorylation and promotes formation of calmodulin complexes; these processes suppress potassium voltage-gated type 7 (Kv7) channels, leading to depolarization and increased nociceptive pain [
17]. In animal models, venom-induced hyperalgesia in rats was reduced by histamine receptor 1 and 2 antagonists [
18,
19]. Collectively, these observations suggest that nociceptive symptoms, as well as itching, should be considered in CU.
Itching and nociceptive signals are typically transmitted through C-fibers [
5]. Several theories have been proposed to explain the relationship between itching and nociceptive pain; one is the intensity theory, which posits that the transition from itch to pain occurs because of increased nociceptor discharge frequency [
4,
20]. In the 3 cases described here, itching consistently preceded nociceptive pain. After nociceptive pain resolved, itching was alleviated but then returned (
Fig. 3) before ultimately resolving. These clinical observations appear consistent with the intensity theory. In addition, the disappearance of nociceptive pain alongside decreasing CU severity during IHC therapy also seems to support this explanation (
Fig. 2).
In a previous report, CU symptoms were schematically classified according to disease severity [
8]. After IHC therapy, symptoms reportedly resolved in parallel with decreasing clinical severity, beginning with angioedema, followed by urticaria, and ultimately itching as severity continued to decline. This pattern of resolution may be related to the intensity of allergic effects. Systemic manifestations, including angioedema, respiratory difficulty, and nociceptive pain, may represent the most severe symptoms, followed by urticaria and then itching as the least severe symptom.
In these patients, nociceptive pain was the first symptom to resolve as CU improved. More broadly, symptom severity may be related to the emergence of nociceptive pain during episodes and as the disease progresses. Thus, these findings suggest that nociceptive pain may reflect a more severe manifestation of CU than urticaria and itching.
To our knowledge, formal reports emphasizing the transition and coexistence of itching and pain in CU have been limited. In addition, the clinical transition between itching and nociceptive pain has not been described in detail. Although this report includes only 3 cases, it describes clinical characteristics of itching and nociceptive pain during symptomatic episodes and the resolution of nociceptive pain as CU severity decreased during IHC therapy. The clinical changes observed during IHC therapy also appear to support the intensity theory regarding the transition from itching to nociceptive pain. Histamine may mediate the resolution of nociceptive pain, based on the effects of histamine receptor 1 blockade and IHC therapy, which has histaminopexy effects.
The primary limitation of this analysis is that, as a descriptive case series involving only 3 patients, the findings cannot be conclusive. In addition, the absence of a control group represents a clear methodological limitation.
In conclusion, nociceptive pain occurred in association with typical urticaria manifestations in 3 patients with CU. Based on the clinical findings reported in this case series, nociceptive pain may represent a more severe manifestation than urticaria and itching. The observed clinical characteristics and response to IHC therapy appear to support the intensity theory regarding the transition from itching to nociceptive pain, which may be mediated by histamine. Pain may represent a type of histamine-mediated syndrome. Further clinical and basic immunological studies are needed.
-
Authors’ contribution
Conceptualization: GN, HK. Data curation: JS. Methodology/formal analysis/validation: YN. Project administration: GN. Funding acquisition: none. Writing–original draft: YN. Writing–review & editing: HK, JS, YN, GN.
-
Conflict of interest
No potential conflicts of interest relevant to this article are reported.
-
Funding
None.
-
Data availability
Not applicable.
-
Acknowledgments
We appreciate all patients who agreed to receive IHC therapy in the interest of advancing medical science and effective treatment. Additionally, we sincerely thank our colleagues and families for their support.
-
Supplementary materials
None.
Fig. 1.Urticaria in a patient with chronic urticaria (case 1).
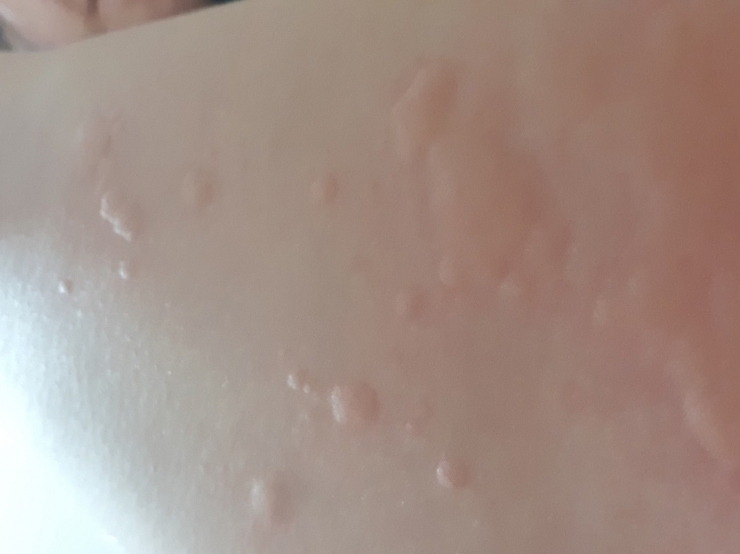
Fig. 2.Changes in nociceptive pain and clinical severity during immunoglobulin/histamine complex (IHC) therapy. (A) Case 1. (B) Case 2. (C) Case 3. Nociceptive pain resolved with decreasing clinical severity of chronic urticaria during IHC therapy. The green arrows in (A), (B), and (C) indicate the points at which nociceptive pain resolved. CSU, chronic spontaneous urticaria; UAS7, urticaria activity score for 7 days; NRS, numeric rating score.
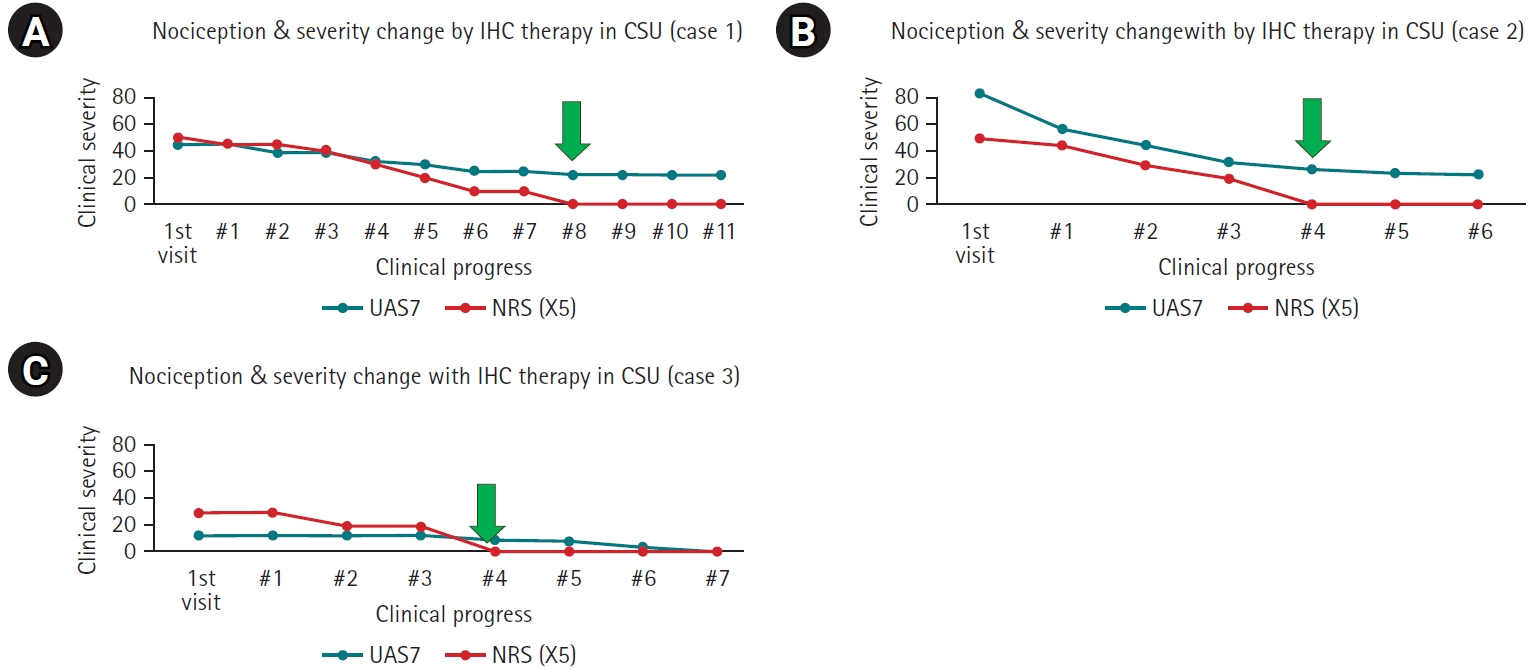
Fig. 3.Schematic of changes during an episode of itching and nociceptive pain symptoms in a representative case (case 1). Severity on the y-axis is shown schematically based on patient descriptions. Nociceptive pain and itching resolved after administration of an antihistamine (histamine receptor 1 blocker).
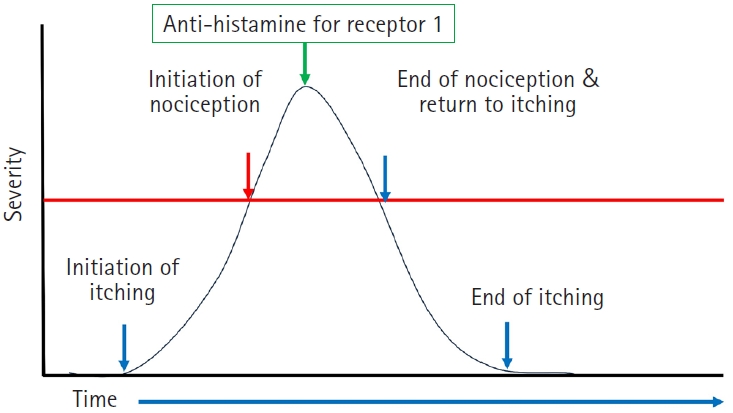
Table 1.Results of basic allergy laboratory tests
|
Case 1 |
Case 2 |
Case 3 |
|
Laboratory tests (normal range, unit) |
|
|
|
|
Blood eosinophil fraction (0%–5%) |
1.7 |
0.2 |
3 |
|
Blood basophil fraction (0.0%–1.0%) |
0.9 |
0.1 |
0.5 |
|
Serum eosinophil cationic protein (0–24 ng/mL) |
48.9 |
16 |
104 |
|
Serum total IgE (≤350 IU/mL) |
52 |
179 |
726 |
|
Serum IgA (70–400 mg/dL) |
271 |
232 |
240 |
|
MAST allergens (IU/mL) |
|
|
|
|
Alternaria alternata
|
<0.35 |
<0.35 |
<0.35 |
|
Aspergillus fumigatus
|
<0.35 |
<0.35 |
<0.35 |
|
Penicillium notatum
|
<0.35 |
<0.35 |
<0.35 |
|
Cladosporium herbarum
|
<0.35 |
<0.35 |
<0.35 |
|
Cockroach |
<0.35 |
<0.35 |
<0.35 |
|
House dust mites |
<0.35 |
<0.35 |
<0.35 |
|
Dermatophagoides pteronyssinus
|
<0.35 |
1.54 |
19.79 |
|
D. farinae
|
<0.35 |
0.65 |
5.37 |
|
Dog |
<0.35 |
<0.35 |
<0.35 |
|
Cat |
<0.35 |
<0.35 |
<0.35 |
|
Birch-Alder mix |
<0.35 |
<0.35 |
9.05 |
|
Mugwort |
<0.35 |
<0.35 |
0.97 |
|
Short Ragweed |
<0.35 |
<0.35 |
0.98 |
|
Sallow willow |
<0.35 |
<0.35 |
<0.35 |
|
Orchard grass |
<0.35 |
<0.35 |
46.56 |
|
Bermuda grass |
<0.35 |
<0.35 |
0.47 |
|
Timothy grass |
<0.35 |
<0.35 |
97.76 |
|
Sweet vernal grass |
<0.35 |
<0.35 |
19.71 |
|
Rye pollen |
<0.35 |
<0.35 |
29.37 |
|
White oak |
<0.35 |
<0.35 |
<0.35 |
|
Japanese cedar |
<0.35 |
<0.35 |
0.38 |
|
Reed |
<0.35 |
<0.35 |
<0.35 |
|
Japanese hop |
<0.35 |
<0.35 |
<0.35 |
|
Acacia |
<0.35 |
<0.35 |
<0.35 |
|
Pine |
<0.35 |
<0.35 |
<0.35 |
|
Poplar mix |
<0.35 |
<0.35 |
<0.35 |
|
Sycamore |
<0.35 |
<0.35 |
<0.35 |
|
Ash mix |
<0.35 |
<0.35 |
0.35 |
|
Oxeye daisy |
<0.35 |
<0.35 |
<0.35 |
|
Dandelion |
<0.35 |
<0.35 |
<0.35 |
|
Russian thistle |
<0.35 |
<0.35 |
<0.35 |
|
Goldenrod |
<0.35 |
<0.35 |
0.95 |
|
Pigweed |
<0.35 |
<0.35 |
0.38 |
|
Crab |
<0.35 |
<0.35 |
<0.35 |
|
Shrimp |
<0.35 |
<0.35 |
<0.35 |
|
Mackerel |
<0.35 |
<0.35 |
<0.35 |
|
Soybean |
<0.35 |
<0.35 |
<0.35 |
|
Hazelnut |
<0.35 |
<0.35 |
0.98 |
|
Peach |
<0.35 |
<0.35 |
<0.35 |
|
Milk |
<0.35 |
<0.35 |
<0.35 |
|
Egg white |
<0.35 |
<0.35 |
<0.35 |
|
Skin prick test (grade) allergens |
|
|
|
|
Alternaria alternata
|
- |
- |
- |
|
Aspergillus fumigatus
|
- |
- |
- |
|
Aspergillus niger
|
- |
- |
- |
|
Candida albicans
|
- |
- |
- |
|
Cladosporium
|
- |
- |
2 |
|
Penicillium chrysogenum
|
- |
- |
- |
|
German cockroach |
- |
2 |
- |
|
D. pteronyssinus
|
- |
3 |
3 |
|
D. farinae
|
- |
3 |
3 |
|
Dog |
- |
- |
- |
|
Cat |
- |
- |
- |
|
Grey alder, silver birch |
- |
- |
2 |
|
Grass mix |
- |
- |
4 |
|
Mugwort |
- |
- |
4 |
|
Short ragweed |
- |
- |
- |
|
Black willow pollen |
- |
- |
- |
|
Orchard |
- |
- |
4 |
|
Bermuda grass |
- |
- |
- |
|
Timothy |
- |
- |
- |
|
English plantain |
- |
- |
- |
|
English ryegrass |
- |
- |
4 |
|
Holm oak |
- |
- |
- |
|
Japanese cedar |
- |
- |
2 |
|
Latex |
- |
- |
- |
|
Milk |
- |
- |
- |
|
Egg |
- |
- |
- |
|
Chicken |
- |
- |
- |
|
Beef |
- |
- |
- |
|
Pork |
- |
- |
- |
|
Cod |
- |
- |
- |
|
Oyster |
- |
- |
- |
|
Salmon |
- |
- |
- |
|
Prawn |
- |
- |
- |
|
Mackerel |
- |
- |
- |
|
Tuna |
- |
- |
- |
|
Almond |
- |
- |
- |
|
Peanut |
- |
- |
- |
|
Bean |
- |
- |
- |
|
Carrot |
- |
- |
- |
|
Cabbage |
- |
- |
- |
|
Walnut |
- |
- |
- |
|
Maize |
- |
- |
- |
|
Peach |
- |
- |
- |
|
Tomato |
- |
- |
- |
|
Black pepper |
- |
- |
- |
|
Spinach |
- |
- |
- |
|
Wheat |
- |
- |
- |
|
Rabbit |
- |
- |
- |
|
Kapok |
- |
- |
- |
|
Hop |
- |
- |
- |
|
F acacia |
- |
- |
- |
|
Pine |
- |
- |
- |
|
Poplar |
- |
- |
- |
References
- 1. Maurer M, Eyerich K, Eyerich S, Ferrer M, Gutermuth J, Hartmann K, Jakob T, Kapp A, Kolkhir P, Larenas-Linnemann D, Park HS, Pejler G, Sánchez-Borges M, Schäkel K, Simon D, Simon HU, Weller K, Zuberbier T, Metz M. Urticaria: collegium internationale allergologicum (CIA) update 2020. Int Arch Allergy Immunol 2020;181:321-333. https://doi.org/10.1159/000507218
- 2. Mizuno M, Fukunaga A, Washio K, Imamura S, Oda Y, Nishigori C. A visual analogue scale for itch and pain in 23 cases of cholinergic urticaria. J Eur Acad Dermatol Venereol 2020;34:e493-e495. https://doi.org/10.1111/jdv.16410
- 3. Grattan CE, Wallington TB, Warin RP, Kennedy CT, Bradfield JW. A serological mediator in chronic idiopathic urticaria: a clinical, immunological and histological evaluation. Br J Dermatol 1986;114:583-590. https://doi.org/10.1111/j.1365-2133.1986.tb04065.x
- 4. Schmelz M. Itch and pain. Neurosci Biobehav Rev 2010;34:171-176. https://doi.org/10.1016/j.neubiorev.2008.12.004
- 5. Schmelz M, Schmidt R, Bickel A, Handwerker HO, Torebjörk HE. Specific C-receptors for itch in human skin. J Neurosci 1997;17:8003-8008. https://doi.org/10.1523/JNEUROSCI.17-20-08003.1997
- 6. Kim JH, Shin IS, Lee YK, Oh HJ, Ban SJ. Improved HPLC method using 2,3-naphthalenedicarboxaldehyde as fluorescent labeling agent for quantification of histamine in human immunoglobulin preparations. Osong Public Health Res Perspect 2011;2:127-134. https://doi.org/10.1016/j.phrp.2011.07.003
- 7. Parrot JL, Laborde C. The histamine-fixing power of blood serum; its modification after an injection of normal human serum or of an azoprotein of histamine. J Physiol (Paris) 1954;46:492-495.
- 8. Kim HS, Noh G. Effects of the immunoglobulin/histamine complex in chronic spontaneous urticaria focusing on remission induction. Allergy 2022;77:2846-2848. https://doi.org/10.1111/all.15381
- 9. Kim HS, Noh G. Induction of remission in chronic urticaria by immunotherapy using immunoglobulin/histamine complex (Histobulin(TM)): a case report. Allergy Asthma Clin Immunol 2021;17:116. https://doi.org/10.1186/s13223-021-00612-8
- 10. Gushchin IS, Luss LV, Il'ina NI, Larina ON, Pakhomova LA. Therapeutic effectiveness of histaglobin preparations in patients with allergic rhinitis and chronic urticaria. Ter Arkh 1999;71:57-62.
- 11. Kulthanan K, Tuchinda P, Chularojanamontri L, Chanyachailert P, Korkij W, Chunharas A, Wananukul S, Limpongsanurak W, Benjaponpitak S, Wisuthsarewong W, Aunhachoke K, Wessagowit V, Chatchatee P, Wattanakrai P, Jirapongsananuruk O, Klaewsongkram J, Noppakun N, Vichyanond P, Suthipinittharm P, Ruxrungtham K, Singalavanija S, Ngamphaiboon J. Clinical practice guideline for diagnosis and management of urticaria. Asian Pac J Allergy Immunol 2016;34:190-200.
- 12. Di Bona D, Nettis E, Bilancia M, Ridolo E, Minenna E, Nizi MC, Albanesi M, Caiaffa MF, Macchia L. Duration of chronic spontaneous urticaria remission after omalizumab discontinuation: a long-term observational study. J Allergy Clin Immunol Pract 2021;9:2482-2485. https://doi.org/10.1016/j.jaip.2021.02.044
- 13. Ito T, Oguro Y, Kakano T. Studies on the production of anti-histamine antibody in the rabbits immunized with “Histobulin”: a new histamine-human-γ-globulin complex developed by authors. Jpn J Allergol 1971;20:285.
- 14. Ishikawa T, Shimada T, Kessoku N, Kiyoi M. Inhibition of rat mast cell degranulation and histamine release by histamine-rat gammaglobulin conjugate. Int Arch Allergy Appl Immunol 1979;59:403-407. https://doi.org/10.1159/000232287
- 15. Ayoub M, Lallouette P, Sütterlin BW, Bessler WG, Huber M, Mittenbühler K. Modulation of the Th1/Th2 bias by an immunoglobulin histamine complex in the ovalbumin allergy mouse model. Int Immunopharmacol 2003;3:523-539. https://doi.org/10.1016/S1567-5769(03)00031-6
- 16. Jariwala SP, Moday H, de Asis ML, Fodeman J, Hudes G, de Vos G, Rosenstreich D. The Urticaria Severity Score: a sensitive questionnaire/index for monitoring response to therapy in patients with chronic urticaria. Ann Allergy Asthma Immunol 2009;102:475-482. https://doi.org/10.1016/S1081-1206(10)60120-2
- 17. Obara I, Telezhkin V, Alrashdi I, Chazot PL. Histamine, histamine receptors, and neuropathic pain relief. Br J Pharmacol 2020;177:580-599. https://doi.org/10.1111/bph.14696
- 18. De Toni LG, Menaldo DL, Cintra AC, Figueiredo MJ, de Souza AR, Maximiano WM, Jamur MC, Souza GE, Sampaio SV. Inflammatory mediators involved in the paw edema and hyperalgesia induced by Batroxase, a metalloproteinase isolated from Bothrops atrox snake venom. Int Immunopharmacol 2015;28:199-207. https://doi.org/10.1016/j.intimp.2015.06.001
- 19. Liu T, Bai ZT, Pang XY, Chai ZF, Jiang F, Ji YH. Degranulation of mast cells and histamine release involved in rat pain-related behaviors and edema induced by scorpion Buthus martensi Karch venom. Eur J Pharmacol 2007;575:46-56. https://doi.org/10.1016/j.ejphar.2007.07.057
- 20. Von Frey M. Zur physiologie der juckempfindung [On the physiology of itching sensation]. Arch Neerland Physiol 1922;7:142-145.