Abstract
Methylenetetrahydrofolate reductase (MTHFR) gene mutations, particularly homozygous mutations, have been associated with a higher incidence of venous thrombosis, coronary heart disease, and obstetric complications. We report the case of a 41-year-old man who presented with multiple vascular thrombotic events over a period of 4–5 years, including deep vein thrombosis with pulmonary thromboembolism, cerebral venous thrombosis, and posterior circulation stroke. The patient was found to have elevated serum homocysteine levels and subsequently underwent genetic testing for MTHFR mutations after other potential prothrombotic conditions were excluded. This case is notable because compound heterozygous mutations of the MTHFR gene (C677T and A1298C) were identified in association with recurrent vascular thrombotic events. Management focused on long-term anticoagulation and supplementation with vitamin B6, vitamin B12, and folic acid.
-
Keywords: Compound heterozygosity; Hyperhomocysteinemia; MTHFR gene; Thrombosis; Case reports
Introduction
Homocysteine is produced during the metabolism of methionine to cysteine. Vitamins B6, B12, and folate play essential roles in homocysteine metabolism through the transsulfuration and remethylation pathways. Transsulfuration requires vitamin B6 as a cofactor, whereas the remethylation of homocysteine—catalyzed by methionine synthase—requires vitamin B12 as a cofactor [
1,
2]. Methylenetetrahydrofolate reductase (
MTHFR) is an enzyme that plays a critical role in folate metabolism and the regulation of homocysteine levels. Reduced
MTHFR enzymatic activity, often secondary to gene mutations, may result in hyperhomocysteinemia and associated vascular thrombotic events [
2,
3]. This case report describes recurrent thrombosis in a patient likely attributable to compound heterozygosity of the
MTHFR gene.
Case presentation
Ethics statement
Written informed consent for publication was obtained, and the patient’s identifying details were anonymized in the manuscript.
Case report
A 41-year-old man with type 2 diabetes mellitus treated with oral hypoglycemic agents presented with right lower limb swelling associated with pain. He had quit smoking 6 months before symptom onset and abstained from alcohol. The patient had no history of recent surgery, trauma, fever, or immobilization. On examination, the right lower limb was edematous and tender, with no skin blisters. Venous Doppler ultrasonography of the right lower limb showed deep venous thrombosis (DVT) involving the right common iliac vein, extending into the right external iliac vein and the deep veins of the entire right lower limb, as well as the great saphenous vein. Computed tomography (CT) venography showed inferior vena cava thrombosis and thrombosis of the right common iliac vein extending to the popliteal, anterior peroneal, and posterior tibial veins, along with the great saphenous vein. The patient was admitted under the vascular
surgery service and started on subcutaneous low-molecular-weight heparin (LMWH) 60 mg twice daily. CT of the thorax showed thrombi in the segmental and subsegmental branches of both lower lobes. The patient subsequently underwent right lower limb catheter-directed thrombolysis with alteplase injection. His serum homocysteine level was elevated at 58.69 μmol/L, representing intermediate hyperhomocysteinemia. Because the patient had been taking an over-the-counter multivitamin supplement containing vitamin B12 and folate, serum vitamin B12 and folate levels were not measured at that time. However, his vitamin B12 level measured at an outside hospital before admission to our center was reportedly >2,000 pg/mL. Bridging to oral anticoagulation was performed, and the international normalized ratio (INR) was monitored with dose adjustments made accordingly. The INR at discharge was 2.64. The patient remained on regular follow-up with the vascular surgery team, and oral anticoagulation with nicoumalone 2 mg once daily was discontinued 1 year after the first event.
Three years later, the patient was again admitted under the general medicine service for evaluation of severe headache. He reported a 3-day history of throbbing holocranial headache with a pain score of 8/10 on the visual analog scale. There were no associated symptoms of fever, nausea, vomiting, blurred vision, loss of consciousness, seizures, or limb weakness. He had no family history of unprovoked thrombosis. On examination, the blood pressure was 130/80 mm Hg, the patient’s pulse rate was 82/min, and he was afebrile. There was no neck stiffness. His Glasgow coma scale score was 15/15, extraocular movements were full, visual acuity was 6/6, and no focal motor deficit or facial asymmetry was noted. Hemogram results showed polycythemia, with a hemoglobin level of 18.4 g/dL and a hematocrit of 52.8%, and the workup performed for cerebral venous thrombosis (CVT) is summarized in
Table 1. Renal and liver function test results were within normal limits.
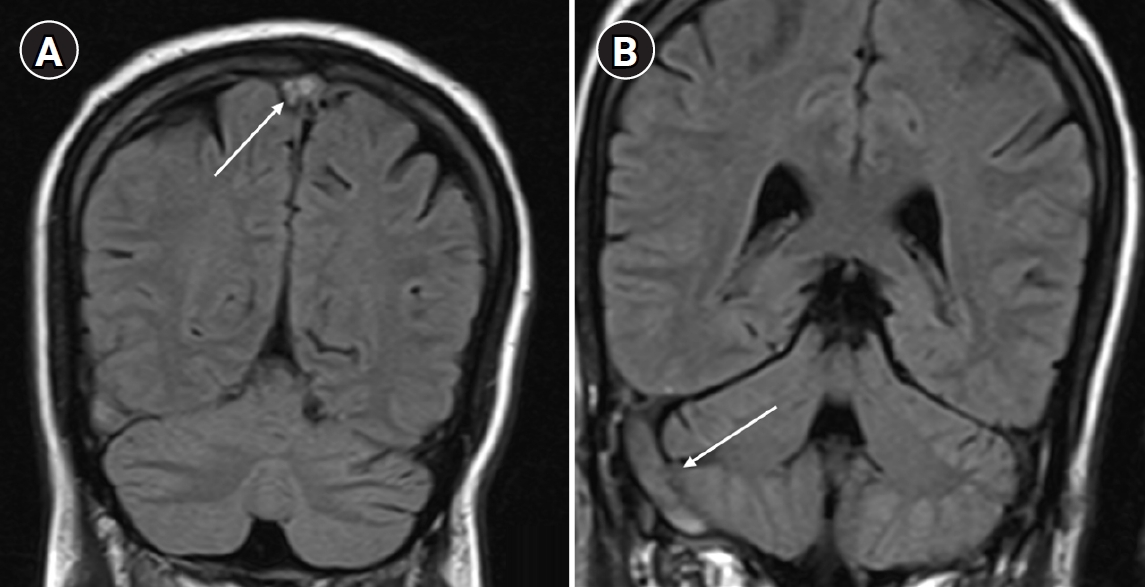
Arterial blood gas analysis performed on room air showed no hypoxemia, with a partial pressure of oxygen of 86 mm Hg. Brain magnetic resonance imaging (MRI) with venography revealed cerebral venous sinus thrombosis involving the superior sagittal sinus, right transverse sinus, and sigmoid sinus (
Fig. 1).
The patient was started on subcutaneous enoxaparin 60 mg twice daily and antiepileptic therapy and was later switched to oral anticoagulation with nicoumalone after a 5-day bridging period with LMWH. Fundus examination showed bilateral papilledema. Given the presence of polycythemia, JAK2 V617F mutation analysis was performed and was negative, and the patient’s erythropoietin level was normal at 7.99 mIU/mL
(reference range, 4.3–29 mIU/mL). Due to the recurrent vascular events, vasculitis and thrombophilia workups were performed and were negative for antinuclear antibodies, antiphospholipid antibody profile, and antineutrophil cytoplasmic antibodies (
Table 1). The patient also had an elevated glycated hemoglobin (HbA1c) level of 11% and dyslipidemia. He was discharged on nicoumalone 2 mg and 3 mg on alternate days, atorvastatin 20 mg, and glipizide 5 mg twice daily. On follow-up, the nicoumalone dose was adjusted to maintain a target INR of 2–3.
Approximately 2 months after the second hospitalization, the patient again presented with giddiness, vomiting, and swaying while walking. MRI of the brain showed a right cerebellar infarct. In addition to oral anticoagulation, single-antiplatelet therapy with aspirin 75 mg and atorvastatin 20 mg was started. The patient reported adherence to oral anticoagulation and had no bleeding symptoms. Echocardiography showed no regional wall motion abnormalities, an ejection fraction of 62%, and normal pulmonary artery pressure. During this hospitalization, HbA1c had decreased from 11% to 9.9%, total cholesterol was 124 mg/dL, high-density lipoprotein cholesterol was 35 mg/dL, and low-density lipoprotein cholesterol was 65 mg/dL. Hemogram results showed a hemoglobin level of 14.3 g/dL and a packed cell volume of 41.6%, suggesting that the previously observed polycythemia was transient. Qualitative polymerase chain reaction (PCR) testing of the MTHFR gene detected heterozygous mutations at both C677T and A1298C. Although diabetes and dyslipidemia may have increased the risk of atherosclerosis and arterial ischemic events, the recurrent venous and arterial thrombotic events in this patient were considered likely attributable to compound heterozygosity (C677T/A1298C genotype) of MTHFR. The patient was switched to a newer oral anticoagulant, dabigatran 150 mg twice daily, and continued on single-antiplatelet and antiepileptic therapy. The switch to the newer oral anticoagulant was made to improve treatment adherence and avoid frequent INR monitoring. On follow-up, the patient reported blurred vision associated with postural variation, especially while walking, that resolved within 2–3 minutes of onset. Ophthalmologic evaluation revealed established papilledema secondary to cortical venous thrombosis. Optical coherence tomography showed thickening of the retinal nerve fiber layer with hemorrhages. The patient was advised to continue lifelong oral anticoagulation and to monitor for bleeding symptoms. He has improved symptomatically, with no active bleeding, and his last recorded INR was 2.21.
Discussion
Homocysteine in blood is predominantly protein-bound (80%), and normal levels range from 5 to 15 μmol/L. Hyperhomocysteinemia can be classified as moderate (15–30 μmol/L), intermediate (30–100 μmol/L), or severe (>100 μmol/L) [
1]. Markedly elevated blood homocysteine levels have atherogenic and prothrombotic properties. Most cases of hyperhomocysteinemia are attributable to vitamin B12 or folate deficiency. Other causes include genetic factors, chronic kidney disease, and smoking. Conditions associated with hyperhomocysteinemia include ischemic heart disease, cerebrovascular disease, venous thromboembolism, obstetric complications, osteoporosis, dementia, and cognitive impairment [
1,
3].
Reduced
MTHFR enzymatic activity can result from polymorphisms in the
MTHFR gene at either the C677T or A1298C locus. Individuals with homozygous mutations carry 2 copies of the same mutant allele, whereas heterozygous individuals carry 1 copy of either C677T or A1298C. Compound heterozygous (double heterozygous) carriers harbor 1 copy each of the C677T and A1298C alleles. In the general population, 60%–70% of individuals may carry 1 of these variants; among them, approximately 2.25% may have compound heterozygous mutations involving both C677T and A1298C, whereas 8.5% may have a homozygous mutation [
4]. Homozygous mutation is more clearly associated with thrombotic events than
MTHFR heterozygosity, for which the association remains a topic of debate. Heterozygous carriers may be at increased risk of recurrent thrombosis in the presence of coexisting risk factors, such as oral contraceptive use or pregnancy. Proposed mechanisms in heterozygous carriers include impaired vascular nitric oxide generation, which may promote oxidative stress and contribute to a prothrombotic milieu [
4].
Naushad et al. [
5] investigated
MTHFR gene polymorphisms using PCR-based restriction fragment length polymorphism analysis in 163 patients with DVT in South India. They reported a 3.5-fold higher risk of DVT among patients with compound heterozygosity (C677T/A1298C genotype) [
5]. This finding is consistent with the recurrent thrombotic events observed in our patient, who was of similar South Indian ethnicity. Ghaznavi et al. [
6] studied
MTHFR gene polymorphisms in 50 Iranian patients with CVT and 50 healthy controls and reported an odds ratio (OR) of 1.35 for patients with the heterozygous C677T genotype and a higher OR of 1.73 in the homozygous 677TT group. However, neither association was
statistically significant, and the study identified
MTHFR gene polymorphism as a significant determinant of homocysteine levels. In a Spanish study of patients with rheumatoid arthritis, the association between
MTHFR gene polymorphism and endothelial dysfunction was evaluated using brachial ultrasonography to assess flow-mediated vasodilatation, with values <7% suggesting endothelial dysfunction. In that study, patients with the heterozygous
MTHFR C677T genotype also demonstrated endothelial dysfunction, with a mean value of 5.9% [
7]. Published data on heterozygous
MTHFR mutations and the risk of vascular thrombosis are summarized in
Table 2 [
5,
6,
8,
9].
The heterozygous A1298C mutation mildly reduces
MTHFR enzymatic activity; however, in the presence of additional risk factors, such as pregnancy or the postpartum state, recurrent thrombosis may occur [
8]. Compound heterozygosity (C677T/A1298C genotype) of the
MTHFR gene may increase the risk of recurrent thrombosis, particularly when additional risk factors are present. It should be noted, however, that compound heterozygosity alone, especially in the absence of other concomitant thrombophilic conditions, has not been conclusively associated with a high thrombotic risk.
In conclusion, this case report highlights a rare presentation of multiple unprovoked thrombotic events despite anticoagulation, likely attributable to compound heterozygosity of the MTHFR gene. However, a multifactorial contribution cannot be excluded in this patient, given the transient nature of the polycythemia, suboptimal glycemic control, and dyslipidemia, all of which may have contributed to large-vessel atherosclerosis. Testing for genetic thrombophilia, including MTHFR polymorphisms, may be worth considering in patients with unexplained recurrent unprovoked thrombotic events.
-
Authors’ contribution
Conceptualization: YSS, VP, DS. Data curation: YSS, VP. Investigation: VP, DS. Methodology: VP, DS. Writing–original draft: YSS, VP. Writing–review & editing: YSS, VP, DS. All the work was done by all authors.
-
Conflict of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Not applicable.
-
Acknowledgments
None.
-
Supplementary materials
None.
Fig. 1.(A) Coronal and sagittal magnetic resonance imaging (MRI) brain images showing thrombosis of the superior sagittal sinus (arrow). (B) MRI brain image showing thrombosis of the right transverse sinus and sigmoid sinus (arrow).
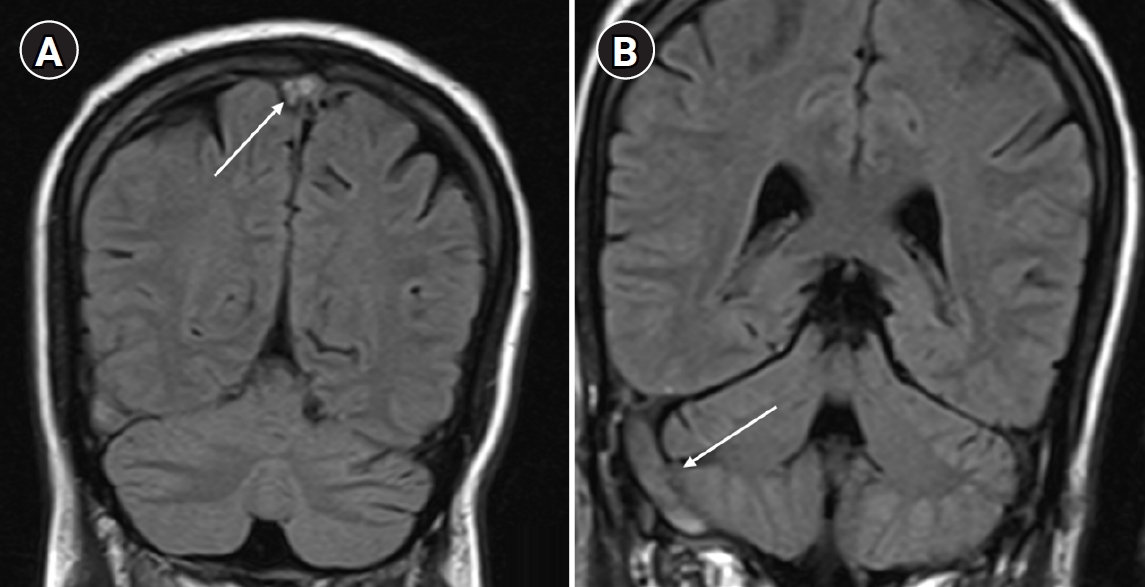
Table 1.Laboratory investigation results
|
Investigations |
Patient value |
Reference value |
|
Hemogram results |
|
|
|
Hemoglobin |
18.1 g/dL |
13–17 g/dL |
|
Hematocrit |
50.7% |
40%–50% |
|
White blood cell count |
8,650 cells/mm3
|
4,000–11,000 cells/mm3
|
|
Platelet count |
255,000/µL |
150,000–450,000/µL |
|
Renal profile |
|
|
|
Blood urea nitrogen |
10 mg/dL |
8–23 mg/dL |
|
Serum creatinine |
0.9 mg/dL |
0.7–1.2 mg/dL |
|
Liver function tests |
|
|
|
Aspartate aminotransferase |
18 IU/L |
<32 IU/L |
|
Alanine aminotransferase |
17 IU/L |
<33 IU/L |
|
Alkaline phosphatase |
113 IU/L |
25–104 IU/L |
|
Total protein |
7.1 g/dL |
6.6–8.7 g/dL |
|
Fasting lipid profile |
|
|
|
Total cholesterol |
212 mg/dL |
<200 mg/dL |
|
Triglycerides |
361 mg/dL |
<150 mg/dL |
|
Low-density lipoprotein cholesterol |
126 mg/dL |
<100 mg/dL |
|
Autoimmune workup |
|
|
|
ANA by immunofluorescence assay |
Negative |
|
|
c-ANCA and p-ANCA |
Negative |
|
|
Anticardiolipin antibodies (IgG and IgM) |
Negative |
|
|
Anti-beta-2 glycoprotein antibodies (IgG and IgM) |
Negative |
- |
|
Lupus anticoagulant |
Not detected |
|
|
Viral markers |
|
|
|
HIV-1/2 with p24 antigen |
Negative |
- |
|
HBsAg |
Negative |
|
|
Anti-HCV |
Negative |
|
Table 2.Published studies on heterozygous MTHFR gene mutations and vascular events
|
Study no. |
Article title |
Reported findings |
|
1. |
H yperhomocysteinemia and the compound heterozygous state for methylene tetrahydrofolate reductase are independent risk factors for deep vein thrombosis among South Indians [5] |
MTHFR genotyping was performed in 163 South Indian patients with DVT. The compound heterozygous C677T/A1298C genotype was identified as an independent risk factor associated with a 3.5-fold higher risk of thrombosis. |
|
2. |
Association study of methylenetetrahydrofolate reductase C677T mutation with cerebral venous thrombosis in an Iranian population [6] |
In this study of 50 Iranian patients with CVT, higher plasma homocysteine levels were observed among patients with MTHFR polymorphisms, and the heterozygous C677T MTHFR genotype was associated with an OR of 1.35 for CVT. However, this association was not statistically significant (95% CI, 0.64–2.84; P=0.56). |
|
3. |
Cerebral vein thrombosis associated with MTHFR A1298C mutation in a young postpartum woman [8] |
A postpartum woman who developed CVT despite anticoagulation was found to have a heterozygous A1298C mutation without a C677T polymorphism. |
|
4. |
Compound heterozygosity for the C677T and A1298C mutations of the MTHFR gene in a case of hyperhomocysteinemia with recurrent deep thrombosis at young age [9] |
A 21-year-old woman with recurrent DVT and cryptogenic cirrhosis was found on evaluation to have compound MTHFR heterozygosity (C677T and A1298C). |
References
- 1. Son P, Lewis L. Hyperhomocysteinemia [Internet]. StatPearls Publishing; 2022 [cited 2025 Oct 10]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK554408/
- 2. Stein JH, McBride PE. Hyperhomocysteinemia and atherosclerotic vascular disease: pathophysiology, screening, and treatment. Arch Intern Med 1998;158:1301-1306. https://doi.org/10.1001/archinte.158.12.1301
- 3. Eichinger S. Homocysteine, vitamin B6 and the risk of recurrent venous thromboembolism. Pathophysiol Haemost Thromb 2003;33:342-344. https://doi.org/10.1159/000083825
- 4. Cole L, Cernasev A, Webb K, Kumar S, Rowe AS. A study of the MTHFR gene prevalence in a rural Tennessee opioid use disorder treatment center population. Int J Environ Res Public Health 2022;19:3255. https://doi.org/10.3390/ijerph19063255
- 5. Naushad S, Jamal NJ, Angalena R, Prasad CK, Devi AR. Hyperhomocysteinemia and the compound heterozygous state for methylene tetrahydrofolate reductase are independent risk factors for deep vein thrombosis among South Indians. Blood Coagul Fibrinolysis 2007;18:113-117. https://doi.org/10.1097/MBC.0b013e3280108e01
- 6. Ghaznavi H, Soheili Z, Samiei S, Soltanpour MS. Association study of methylenetetrahydrofolate reductase C677T mutation with cerebral venous thrombosis in an Iranian population. Blood Coagul Fibrinolysis 2015;26:869-873. https://doi.org/10.1097/MBC.0000000000000292
- 7. Palomino-Morales R, Gonzalez-Juanatey C, Vazquez-Rodriguez TR, Rodriguez L, Miranda-Filloy JA, Fernandez-Gutierrez B, Llorca J, Martin J, Gonzalez-Gay MA. A1298C polymorphism in the MTHFR gene predisposes to cardiovascular risk in rheumatoid arthritis. Arthritis Res Ther 2010;12:R71. https://doi.org/10.1186/ar2989
- 8. Bostan A, Țăpoi LC, Barcan MN, Florea L. Cerebral vein thrombosis associated with MTHFR A1289C mutation gene in a young postpartum woman. Arch Clin Cases 2019;6:26-30. https://doi.org/10.22551/2019.22.0601.10150
- 9. Jurcuţ R, Pop I, Coriu D, Grasu M, Zilişteanu D, Giuşcă S, Ginghină C. Compound heterozygosity for the C677T and A1298C mutations of the MTHFR gene in a case of hyperhomocysteinemia with recurrent deep thrombosis at young age. Rom J Intern Med 2008;46:255-259.