Abstract
We present a rare case of a 68-year-old woman who experienced dizziness, nausea, vomiting, and imbalance for 2 months. Imaging studies, including magnetic resonance imaging, magnetic resonance angiography, and digital subtraction angiography, revealed an intrasellar course of the persistent trigeminal artery (PTA) with atherosclerotic changes. Additionally, hypoplasia of the basilar artery and left vertebral artery was observed. The anatomically rare intrasellar PTA, combined with atherosclerosis and vertebrobasilar hypoplasia, likely contributed to compromised posterior circulation and the patient’s symptoms. This case highlights the importance of considering vascular anomalies and the associated pathological changes in patients with otherwise unexplained posterior circulation symptoms.
-
Keywords: Intrasellar persistent trigeminal artery; Persistent trigeminal artery; Atherosclerosis; Vertebrobasilar insufficiency; Vertebrobasilar hypoplasia; Case reports
Introduction
Persistent trigeminal artery (PTA) is the most common carotid–vertebrobasilar anastomosis, with a reported incidence of 0.1%–0.6% [
1-
3]. It typically arises from the posterolateral aspect of the cavernous internal carotid artery (ICA), traverses Meckel’s cave, and joins the basilar artery, providing a direct connection between the anterior and posterior circulations. Although PTA is usually asymptomatic, it may be associated with vascular anomalies such as hypoplasia, aneurysms, or atherosclerotic changes, which can alter hemodynamics and produce posterior circulation symptoms [
4-
7]. An intrasellar course of the PTA is a very rare anatomical variant that can pose diagnostic challenges and may have clinical significance when coexisting vascular pathology is present. This case report describes an unusual presentation of symptomatic intrasellar PTA with atherosclerotic narrowing associated with dizziness.
Case presentation
This retrospective study was approved by the institutional review board of Ewha Womans University Seoul Hospital, Korea (2025-06-090-001), and the requirement for informed consent was waived.
A 68-year-old woman with hypertension and dyslipidemia visited the outpatient clinic of our hospital, reporting dizziness, nausea, vomiting, and imbalance that had persisted for 2 months. The dizziness was non-rotatory and was intermittently accompanied by a subjective sense of gait instability.
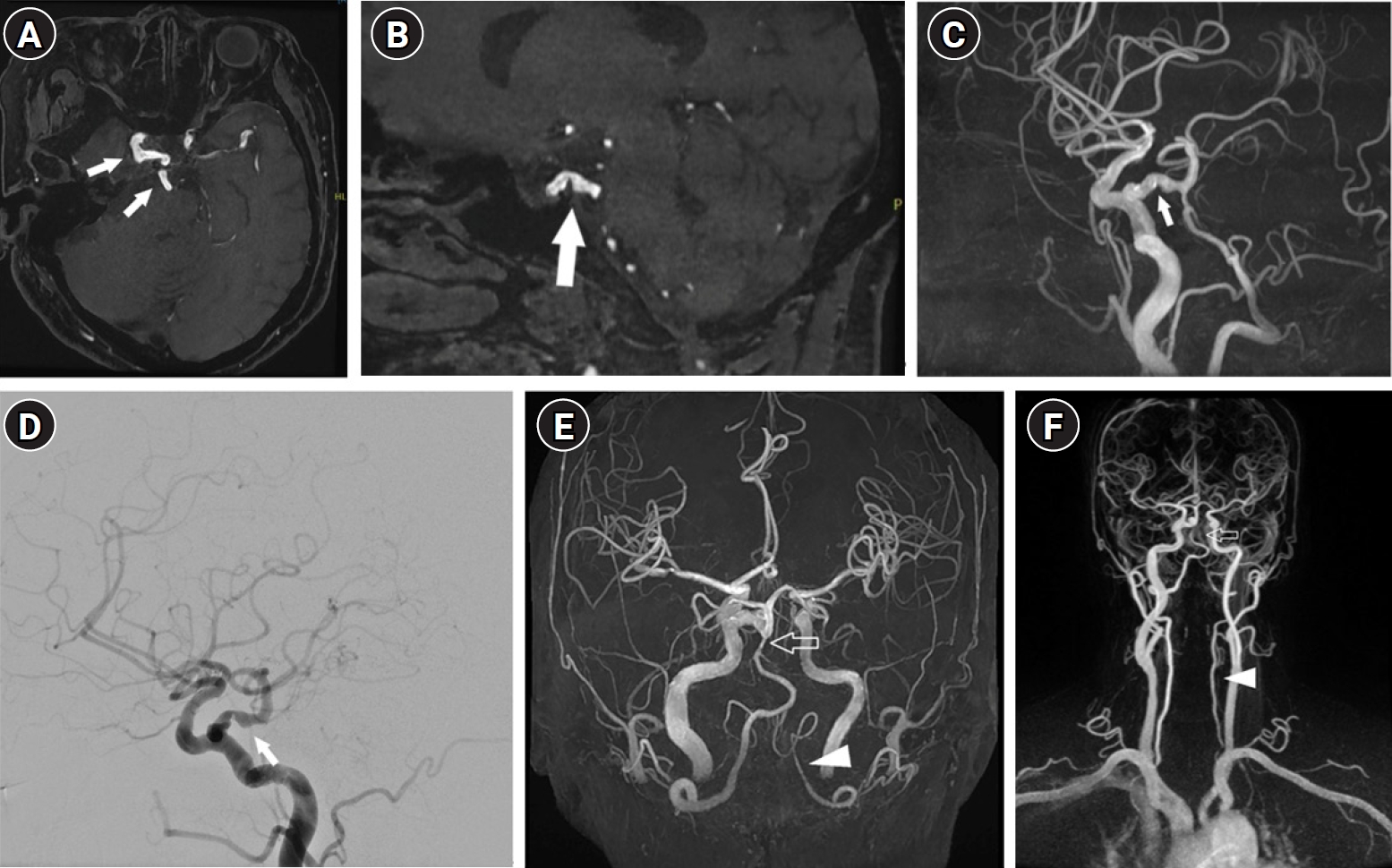
Neurological examination was largely unremarkable except for mild gait instability. Contrast-enhanced brain magnetic resonance imaging revealed no definite acute infarction or space-occupying lesion. Magnetic resonance angiography and digital subtraction angiography (DSA) showed a right intrasellar PTA arising from the cavernous segment of the right ICA and passing through the sellar fossa to join a hypoplastic basilar artery (
Fig. 1). DSA also revealed luminal narrowing and irregularity of the right PTA, findings consistent with atherosclerosis (
Fig. 1). Additionally, hypoplasia of the basilar artery and left vertebral artery was identified (
Fig. 1). This case corresponds to Saltzman type I PTA, which typically connects the ICA to the basilar artery and compensates for vertebral artery hypoplasia. The anatomical and hemodynamic features of this rare PTA variant likely contributed to compromised posterior circulation and the patient’s symptoms. Aside from ongoing medication for hypertension and dyslipidemia, no additional specific treatment was administered. Follow-up magnetic resonance imaging demonstrated no significant interval change.
This case highlights the potential clinical significance of an intrasellar PTA in posterior circulation insufficiency, particularly when accompanied by vascular anomalies and atherosclerotic changes.
Discussion
An intrasellar course of the PTA is a very rare anatomical variant, and symptomatic presentation with superimposed atherosclerosis is even rarer. Previous reports of vertebrobasilar insufficiency associated with PTA have predominantly implicated stenosis of the ICA as the underlying cause [
8-
10]. Some reports have described PTA involvement in the setting of ICA stenosis [
8,
10]. In contrast, our case demonstrated isolated atherosclerotic narrowing of the PTA without concurrent ICA stenosis. PTA is often considered an incidental finding; however, in this patient, it served as the dominant supply to the posterior circulation. Therefore, atherosclerotic compromise of the PTA appeared to have direct symptomatic consequences. To our knowledge, this is the first reported case of intrasellar PTA stenosis resulting in vertebrobasilar insufficiency in the absence of ICA stenosis.
To date, no established theory or direct evidence has clearly explained the mechanism by which an intrasellar PTA develops atherosclerosis. However, its unique anatomical location and hemodynamic characteristics suggest several plausible contributing factors. The intrasellar course carries the artery through the sellar fossa, an anatomically constrained space that may subject the vessel to abnormal mechanical forces [
11,
12]. Mechanical compression within the narrow confines of the sellar fossa could disrupt normal flow patterns in the PTA. Such altered hemodynamics, characterized by low wall shear stress and increased turbulence, are well-recognized contributors to endothelial dysfunction and subsequent atherogenesis [
13-
16]. In addition, the PTA’s acute angulation at its origin and termination may further increase localized stress on the vessel wall. Repeated pulsatile contact with surrounding bony structures could cause microvascular trauma and provoke chronic inflammatory responses, thereby accelerating atherosclerotic plaque formation. In our case, vertebrobasilar hypoplasia was also present. Saltzman type I PTA typically connects the ICA to the basilar artery to compensate for vertebral artery hypoplasia, as observed in this case. The coexisting left vertebral artery hypoplasia reduced posterior circulation reserve, rendering the PTA a critical conduit for posterior cerebral perfusion. Any stenosis or other flow-limiting pathology in this vessel could therefore lead to posterior circulation insufficiency, manifesting as dizziness or even transient ischemic symptoms. Nevertheless, these proposed mechanisms remain speculative because direct histopathological evidence or large-scale imaging studies confirming atherosclerosis specifically within an intrasellar PTA are lacking. Further research is needed to clarify the vascular pathology associated with this rare anatomical variant.
Saltzman first described the classification of PTA, dividing it into 3 types on the basis of angiographic features [
17]. In Saltzman type I, the PTA joins the basilar artery between the origins of the superior cerebellar artery (SCA) and the anterior inferior cerebellar artery (AICA). In this variant, the proximal basilar artery and posterior communicating artery (PComA) are typically hypoplastic, and the PTA supplies both posterior cerebral arteries (PCAs) and SCAs. In Saltzman type II, the PTA connects with the basilar artery at a level superior to the origin of the SCAs, whereas the PComAs remain intact and supply the PCAs. Type III has traditionally been regarded as a combination of types I and II. Subsequently, Ali et al. [
18] refined the Saltzman classification by incorporating additional PTA variants into type III, characterized by the absence of a basilar artery connection. In these variants, the PTA arises from the ICA and terminates directly as the SCA (type IIIa), AICA (type IIIb), or posterior inferior cerebellar artery (type IIIc), without interposition of the basilar artery. Our case corresponds to Saltzman type I PTA, which typically connects the ICA to the basilar artery and compensates for hypoplastic basilar and vertebral arteries. Several reports have described PTA associated with posterior circulation symptoms [
4-
6,
9,
19]. Ferreira et al. [
19] documented a case of Saltzman type I PTA that resulted in an acute ischemic event because of decreased vascular supply to the posterior fossa. Other studies have reported posterior territory infarction due to embolism from ICA stenosis or dissection via the PTA [
20,
21]. Battista et al. [
9] described extensive thrombosis of the cavernous ICA and PTA secondary to proximal ICA dissection, in which the PTA, normally serving as a collateral route, became the primary pathway for the posterior circulation. In these cases, ICA occlusion compromised this essential collateral channel, leading to ischemic events in both circulatory territories. However, these previously reported cases were largely attributable to secondary vascular conditions such as ICA stenosis or vertebral artery hypoplasia, in which the PTA functioned as a compensatory route [
4,
7,
22]. Lee and Kelly [
12] described an intrasellar PTA associated with a pituitary adenoma, but no evidence of atherosclerosis was identified. To date, no previous case has clearly documented atherosclerotic changes within an intrasellar PTA as the direct cause of symptoms. Our case is the first to describe symptomatic intrasellar PTA with angiographically confirmed atherosclerotic narrowing, providing novel insight into the potential for primary vascular pathology within this embryonic remnant to produce clinically significant posterior circulation insufficiency.
In summary, this case highlights a potential association between the intrasellar course of the PTA and the development of atherosclerotic changes, likely driven by altered hemodynamics. The anatomical constraints and resultant turbulent flow in this rare variant may predispose the vessel wall to early pathological changes. In patients presenting with posterior circulation symptoms such as dizziness, an intrasellar PTA should be considered in the differential diagnosis. Moreover, PTA-associated atherosclerosis in the setting of vertebrobasilar hypoplasia may further exacerbate hemodynamic compromise and contribute to symptomatic posterior circulation insufficiency. Comprehensive vascular imaging is therefore essential for accurate diagnosis and appropriate management planning.
-
Authors’ contribution
Conceptualization: BL. Data curation: JB, BL. Formal analysis: JB, BL. Methodology: JB, BL. Validation: JB, BL. Investigation: JB, BL. Supervision: BL. Writing–original draft: JB, BL. Writing–review & editing: JB, BL.
-
Conflict of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Not applicable.
-
Acknowledgments
None.
-
Supplementary materials
None.
Fig. 1.(A, B) Axial and sagittal magnetic resonance angiography (MRA) time-of-flight (TOF) source images show the intrasellar persistent trigeminal artery (PTA) (arrow) arising from the right cavernous internal carotid artery (ICA) and passing through the sellar fossa. (C, D) Brain MRA maximum intensity projection (MIP) and digital subtraction angiography show arterial irregularity with luminal narrowing of the PTA. (E) Brain MRA MIP and (F) neck MRA show hypoplasia of the basilar artery (empty arrow) and vertebral artery (arrowhead), without severe steno-occlusion or dissection of the proximal ICA.
References
- 1. Eadie MJ, Jamieson KG, Lennon EA. Persisting carotid-basilar anastomosis. J Neurol Sci 1964;1:501-511. https://doi.org/10.1016/0022-510x(64)90170-4
- 2. Fields WS. The significance of persistent trigeminal artery: carotid-basilar anastomosis. Radiology 1968;91:1095-1101.
- 3. Bechri H, Louraoui SM, Fikri M, El Fatemi N, El Maaqili MR, El Abbadi N. Persistence of a trigeminal artery associated with a posterior meningeal artery aneurysm: case report and literature review. J Surg Case Rep 2020;2020:rjz389. https://doi.org/10.1093/jscr/rjz389
- 4. Engelhardt J, El Hage G, Bojanowski MW. Persistent trigeminal artery as collateral circulation in ischemic stroke. World Neurosurg 2021;148:67-69. https://doi.org/10.1016/j.wneu.2021.01.034
- 5. Momma F, Ohara S, Ohyama T. Persistent trigeminal artery associated with brainstem infarct: case report. Neurol Med Chir (Tokyo) 1992;32:289-291. https://doi.org/10.2176/nmc.32.289
- 6. Tyagi G, Sadashiva N, Konar S, Aravinda HR, Saini J, Shukla D, Devi BI. Persistent trigeminal artery: neuroanatomic and clinical relevance. World Neurosurg 2020;134:e214-e223. https://doi.org/10.1016/j.wneu.2019.10.025
- 7. Wang Y, Yu J. Clinical importance of the persistent primitive trigeminal artery in vascular lesions and its role in endovascular treatment. Front Neurol 2022;13:928608. https://doi.org/10.3389/fneur.2022.928608
- 8. Hirashima Y, Endo S, Koshu K, Takaku A. Vertebrobasilar insufficiency in a patient with persistent trigeminal artery and stenosis of the ipsilateral carotid bifurcation: case report. Neurol Med Chir (Tokyo) 1988;28:584-587. https://doi.org/10.2176/nmc.28.584
- 9. Battista RA, Kwartler JA, Martinez DM. Persistent trigeminal artery as a cause of dizziness. Ear Nose Throat J 1997;76:43-45. https://doi.org/10.1177/014556139707600112
- 10. Park YH, Jung KH, Roh JK. Vertebrobasilar insufficiency by persistent trigeminal artery stenosis. Neurol Asia 2013;18:311-313.
- 11. Kim MS, Hur JW, Lee JW, Lee HK. Persistent trigeminal artery detected by conventional angiography and magnetic resonance angiography. J Korean Neurosurg Soc 2005;37:101-104.
- 12. Lee KS, Kelly DL. Intrasellar persistent trigeminal artery associated with a pituitary adenoma: case report. J Neurosurg 1989;70:271-273. https://doi.org/10.3171/jns.1989.70.2.0271
- 13. Yamashiro Y, Yanagisawa H. The molecular mechanism of mechanotransduction in vascular homeostasis and disease. Clin Sci (Lond) 2020;134:2399-2418. https://doi.org/10.1042/CS20190488
- 14. Peng Z, Shu B, Zhang Y, Wang M. Endothelial response to pathophysiological stress. Arterioscler Thromb Vasc Biol 2019;39:e233-e243. https://doi.org/10.1161/ATVBAHA.119.312580
- 15. Cheng H, Zhong W, Wang L, Zhang Q, Ma X, Wang Y, Wang S, He C, Wei Q, Fu C. Effects of shear stress on vascular endothelial functions in atherosclerosis and potential therapeutic approaches. Biomed Pharmacother 2023;158:114198. https://doi.org/10.1016/j.biopha.2022.114198
- 16. Li J, Fang Y, Wu D. Mechanical forces and metabolic changes cooperate to drive cellular memory and endothelial phenotypes. Curr Top Membr 2021;87:199-253. https://doi.org/10.1016/bs.ctm.2021.07.003
- 17. Saltzman GF. Patent primitive trigeminal artery studied by cerebral angiography. Acta Radiol (Stockh) 1959;51:329-336. https://doi.org/10.3109/00016925909171103
- 18. Ali S, Radaideh MM, Shaibani A, Russell EJ, Walker MT. Persistent trigeminal artery terminating in the posterior inferior cerebellar artery: case report. Neurosurgery 2008;62:E746-E748. https://doi.org/10.1227/01.neu.0000317327.17225.f8
- 19. Ferreira A, Coelho PS, Cruz VT. Persistent trigeminal artery in a patient with posterior circulation stroke treated with rt-PA: case report. BMC Neurol 2019;19:257. https://doi.org/10.1186/s12883-019-1492-2
- 20. Foerch C, Berkefeld J, Halbsguth A, Ziemann U, Neumann-Haefelin T. Brain stem infarction caused by proximal internal carotid artery stenosis in a patient with a persisting primitive trigeminal artery. Cerebrovasc Dis 2006;22:200-202. https://doi.org/10.1159/000093808
- 21. Iancu D, Anxionnat R, Bracard S. Brainstem infarction in a patient with internal carotid dissection and persistent trigeminal artery: a case report. BMC Med Imaging 2010;10:14. https://doi.org/10.1186/1471-2342-10-14
- 22. Lyu DP, Wang Y, Wang K, Yao M, Wu YF, Zhou ZH. Acute cerebral infarction in a patient with persistent trigeminal artery and homolateral hypoplasia of internal carotid artery distal anastomosis: a case report and a mini review of the literature. J Stroke Cerebrovasc Dis 2019;28:104388. https://doi.org/10.1016/j.jstrokecerebrovasdis.2019.104388
Citations
Citations to this article as recorded by